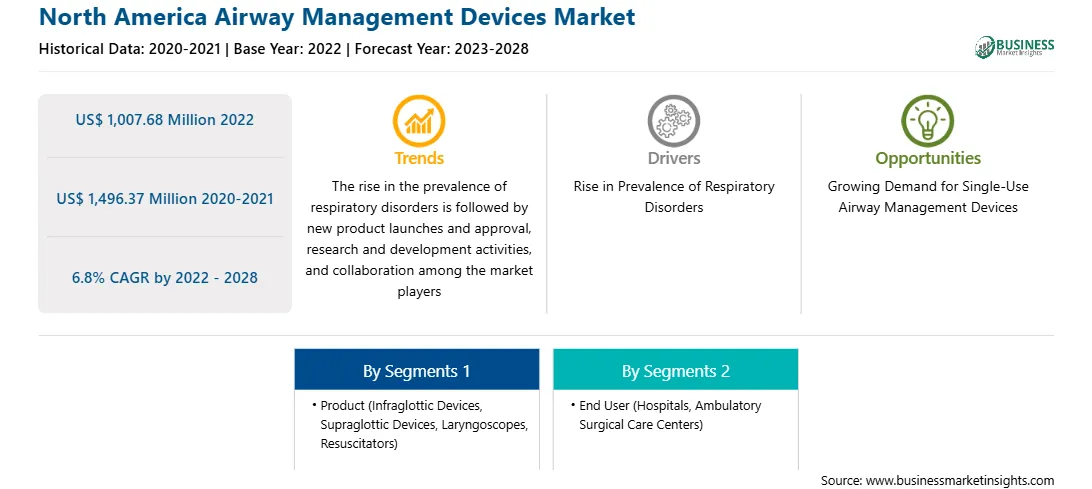
The North America airway management devices market size is expected to reach US$ 4,435.5 million by 2033 from US$ 2,847.5 million in 2025. The market is estimated to record a CAGR of 5.8% from 2026 to 2033.
The North America airway management devices market is considered to be an established market that continues to evolve, characterized by a heavy focus on clinical safety in terms of patient well-being and efficient respiratory support. There has been consistent demand for airway management products in hospitals, ambulatory surgery centers, and emergency medical services within the region owing to the prevalent nature of respiratory diseases, surgeries, and critical care admissions. The rising use of technologically sophisticated equipment like video laryngoscopes and single-use supraglottic airway devices underscores this trend. Infection control and the use of single-use devices have become top priorities among North American healthcare organizations due to the increasing importance of infection control following global healthcare emergencies. As a result, more disposable airway management products are being purchased to minimize the risk of cross-contamination in clinical settings. Also, training and simulation-based learning for clinicians on the use of these devices have been shown to boost the market’s development. There are also various limitations that the market encounters. First, the cost of advanced airway management solutions may restrict their use in smaller healthcare establishments located in rural regions. Second, the existence of strict regulations may also pose a challenge by slowing down the approval process of products and delaying their introduction to the market. Nevertheless, institutional backing, high healthcare expenditure, and quick adoption of new technology make North America an important regional market.
Key segments that contributed to the derivation of the airway management devices market analysis are product type, application, patient type, and end user.
A marked increase in cases of respiratory diseases has been recorded in North America due to reasons like pollution, smoking, occupation, and aging of the population. In the United States alone, the rate of asthma and chronic obstructive pulmonary disease has reached alarming levels with many people suffering from these conditions. The result of this is an ever-growing number of patients and hence a corresponding increase in the need for airway management devices.
Respiratory illnesses in North America also feature rising complexity due to an increase in the number of patients who experience other complications, including obesity, heart problems, and diabetes. These factors tend to make airway management complicated, thus requiring better equipment. Consequently, hospitals are increasingly purchasing high-quality technology such as video-guided intubation and supraglottic airway devices to enhance accuracy and patient safety during the procedures.
Moreover, seasonal outbreaks of respiratory diseases as well as other public health issues have highlighted the need for preparedness in the management of airways. Healthcare organizations in the region are making it a priority to maintain a rapid response system by stockpiling crucial airway tools. As a result, procurement practices have become more structured, with suppliers establishing long-lasting partnerships with hospitals in North America.
Even though North America may seem like a developed market, the growth in some of the developing segments, including outpatient clinics, ambulatory surgery centers, and home healthcare facilities, is noteworthy. Health care financing is focusing more on rural and disadvantaged regions in the US and parts of Canada, and this is spurring the growth of the market by driving demand for portable airway management products that offer an affordable solution.
The trend towards value-based care models is driving healthcare service providers to embrace devices that enhance efficiency while minimizing hospitalization. There is an increasing uptake of devices that facilitate fast recovery, eliminate risks, and facilitate non-invasive procedures. This is especially important in the outpatient surgery environment, where speedy patient turnover is a critical requirement for the operations of such facilities.
Additionally, the high pace of development of telemedicine and remote patient monitoring services in North America is creating opportunities for the innovation and application of airway management devices. Devices with smart and connected functions that help the clinician to monitor and manage their patients from a distance are currently being favored. The integration of technology and respiratory management in the healthcare space creates new opportunities, particularly within the home therapy segment, making emerging healthcare segments a vital part of the market’s growth in the region.
The North America airway management devices market is experiencing steady growth, with market size and share analysis reflecting evolving treatment preferences and competitive dynamics among key players. The report evaluates important subsegments categorized within product type, application, patient type, and end user, highlighting their respective contributions to overall market performance.
By product type, the infraglottic devices subsegment dominated the market in 2025. In anesthesia practice, infraglottic devices provide protection against aspiration and enable precise airway management, driving higher adoption in operating rooms and critical care settings compared to supraglottic devices.
Based on application, the anesthesia subsegment dominated the market in 2025 due to the need for definitive airway control across a wide range of surgical procedures, from routine elective surgeries to complex interventions. Because infraglottic devices are essential for intubation during anesthesia, this segment experiences consistent, high-volume usage, making it the largest segment compared to emergency or general critical care.
In terms of patient type, the adult subsegment dominated the market in 2025. Most procedural training, device designs, and usage guidelines are optimized for adult anatomy and usage scenarios, reinforcing higher market volume.
Based on end user, the hospital subsegment dominated the market in 2025, as hospitals remain the central hub for airway management, handling scheduled surgeries, emergency admissions, ICU care, and trauma cases. Their larger budgets, need for extensive airway inventories, and availability of multidisciplinary professionals make them the largest purchasers compared to outpatient centers or EMS, where volume and purchasing power are comparatively lower.
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 2,847.5 Million |
| Market Size by 2033 | US$ 4,435.5Million |
| CAGR (2026 - 2033) | 5.8% |
| Historical Data | 2022-2024 |
| Forecast period | 2026-2033 |
| Segments Covered | By Product Type
|
|
Regions and Countries Covered
| |
| North America | US, Canada, Mexico |
| Market leaders and key company profiles |
|
The "North America Airway management devices market Size and Forecast (2022–2033)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the North America airway management devices market report is divided into: The US, Canada, and Mexico. The US held the largest share in 2025.
The US represents the dominant market within North America, owing to its well-established health care infrastructure, along with its early adaptability towards innovations in the medical technology segment. There is substantial demand in the US for cutting-edge airway management products within numerous applications, such as emergency departments, intensive care units, and ambulatory surgery centers. The emphasis on enhancing patient outcomes while minimizing airway risks has contributed toward the use of video-assisted and precisely guided airway instruments. There is a clear tendency towards adopting airway management protocols in tandem with the use of digital healthcare solutions, thus facilitating monitoring and documentation of the process. There are many academic medical centers and large hospital chains conducting clinical studies and implementing pilot projects. The other important aspect to mention is that there is a strong presence of medical device manufacturers and product launch events. Another asset the U.S. market offers is its robust training framework, including specialized simulators which allow professionals to improve their skills in airway management. On the downside, one should acknowledge the high prices for technology, as well as the complex reimbursement policy, which could create problems for smaller healthcare facilities. Nevertheless, despite the aforementioned limitations, the country remains the leading player in terms of regional market share thanks to technological advantages, expertise, and patient-centered approach to care.
The North America airway management devices market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the North America Airway management devices market are:
The North America airway management devices market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the North America Airway management devices market are:
The North America Airway Management Devices Market is valued at US$ 2,847.5 Million in 2025, it is projected to reach US$ 4,435.5Million by 2033.
As per our report North America Airway Management Devices Market, the market size is valued at US$ 2,847.5 Million in 2025, projecting it to reach US$ 4,435.5Million by 2033. This translates to a CAGR of approximately 5.8% during the forecast period.
The North America Airway Management Devices Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Airway Management Devices Market report:
The North America Airway Management Devices Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Airway Management Devices Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Airway Management Devices Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)