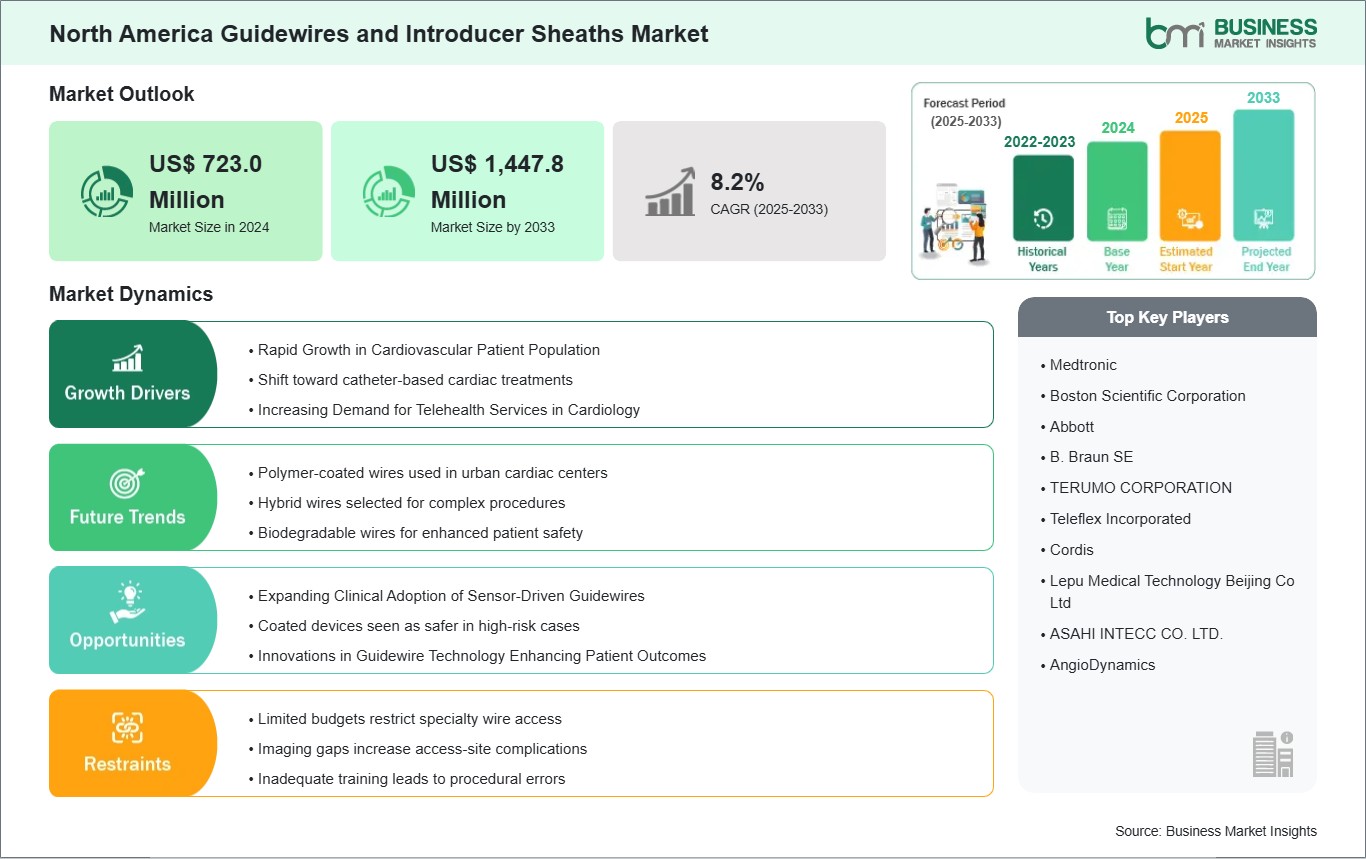
The North America guidewires and introducer sheaths market size is expected to reach US$ 1,447.8 million by 2033 from US$ 723.0 million in 2024. The market is estimated to record a CAGR of 8.2% from 2025 to 2033.
The North America guidewires and introducer sheaths market is defined by advanced hospital infrastructure, strong reimbursement frameworks, and a culture of evidence‑based adoption. Hospitals across the US and Canada are early adopters of minimally invasive cardiovascular and peripheral procedures, supported by robust imaging systems and hybrid operating rooms. A key driver is the region's emphasis on patient safety and efficiency. Clinicians prioritize guidewires with predictable tactile feedback and introducer sheaths engineered for smooth insertion, reliable hemostasis, and rapid catheter exchanges. Procurement is shaped by group purchasing organizations (GPOs), integrated delivery networks (IDNs), and hospital consortia, which negotiate long‑term contracts and favor vendors that demonstrate consistent lot quality, transparent post‑market surveillance, and sustainability in packaging. Another distinctive factor is the integration of digital health and electronic medical records, where consumables are tracked against patient outcomes and replenishment cycles.
Market restraints include strict cost‑effectiveness evaluations, competitive price pressures from bulk tenders, and regulatory requirements under the FDA and Health Canada that extend product launch timelines. Despite these challenges, North America benefits from strong government funding, private sector investment, and academic hospitals that pilot new technologies and generate clinical data for broader adoption. Hospitals remain the dominant end users, while specialized cardiovascular centers and diagnostic networks contribute by standardizing imaging‑led triage into interventional pathways. Vendors that align with GPO procurement models, sustainability initiatives, and evidence‑driven positioning are best placed to sustain growth in this sophisticated market.
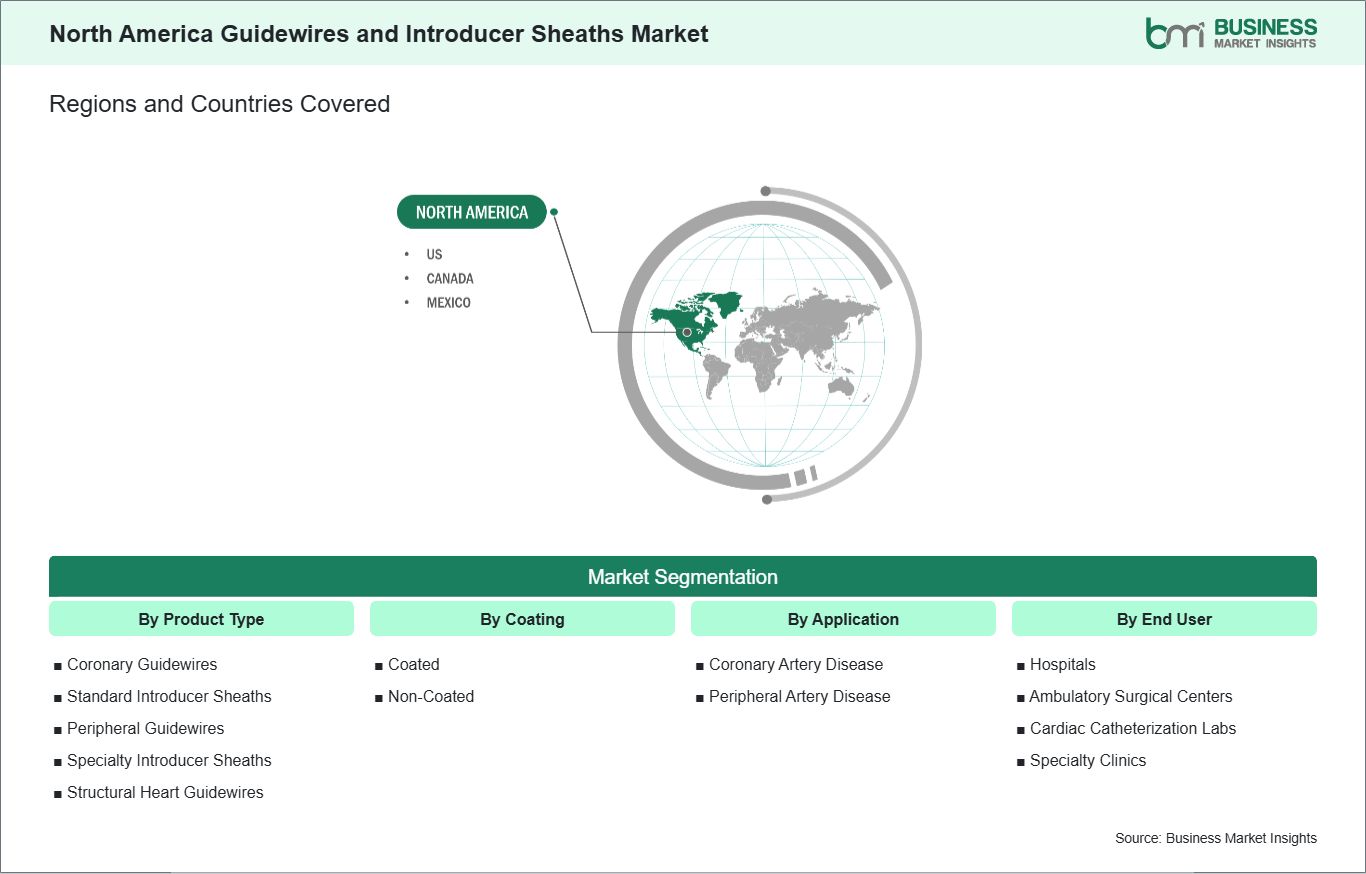
Key segments that contributed to the derivation of the North America guidewires and introducer sheaths market analysis are product type, coating, application, and end user.
North America continues to face one of the world's highest burdens of cardiovascular disease, driven by aging populations, obesity, diabetes, and sedentary lifestyles. In the US, heart disease remains the leading cause of death, while Canada is seeing a rising incidence of coronary artery disease and heart failure among people over 60. These trends translate into millions of diagnostic angiographies, percutaneous coronary interventions, and peripheral vascular procedures each year, creating sustained and large-scale demand for guidewires and introducer sheaths across hospitals and outpatient cardiac centers.
The region's highly developed healthcare infrastructure supports a very high volume of interventional cardiology procedures. Large hospital networks and specialized cardiac institutes in cities such as Houston, Boston, Toronto, and Los Angeles operate multiple catheterization laboratories that perform complex coronary, neurovascular, and structural heart interventions daily. These advanced procedures rely on a wide range of specialized guidewires and introducer sheaths designed for different vessel sizes, lesion types, and access routes, making these devices essential consumables within North American cath labs.
The shift toward minimally invasive treatment pathways is accelerating across North America as payers and providers aim to reduce hospital stays and overall healthcare costs. Procedures such as transcatheter aortic valve replacement (TAVR), endovascular aneurysm repair, and percutaneous coronary intervention are preferred over open surgery. Each of these therapies requires precise vascular access and navigation, increasing the utilization of high-quality guidewires and introducer sheaths. As cardiovascular disease remains a dominant healthcare challenge, these trends ensure strong, long-term market growth.
North America is the global leader in the adoption of sensor-enabled guidewires, driven by its strong innovation ecosystem and widespread acceptance of advanced interventional technologies. Pressure and flow-sensing guidewires are now commonly used in US and Canadian cath labs to perform fractional flow reserve (FFR) and other physiologic assessments during coronary angiography. These measurements help cardiologists determine whether a blockage truly requires stenting, improving patient outcomes and reducing unnecessary interventions.
Major academic medical centers and large hospital systems, such as those in New York, Cleveland, and Vancouver, have incorporated sensor-enabled guidewires into standard clinical protocols for complex coronary and peripheral artery disease. Their ability to provide real-time, objective data supports more precise decision-making, especially in patients with multi-vessel disease or ambiguous lesions. This technology also aligns with the region's growing focus on value-based care, where better outcomes and efficient use of resources are rewarded.
The market is strengthened by the presence of leading medical device manufacturers and a robust clinical research environment in North America. Continuous innovation, rapid regulatory approvals, and extensive physician training programs accelerate the uptake of next-generation sensor-enabled guidewires. As healthcare providers rely on data-driven insights to guide interventional strategies, these advanced devices are becoming a standard of care, reinforcing North America's dominant position in the global guidewires and introducer sheaths market.
The North America Guidewires and Introducer Sheaths market demonstrates steady growth, with size and share analysis highlighting evolving trends and competitive dynamics among key players. The report further examines subsegments categorized within product type, coating, application, and end user, offering insights into their contribution to overall market performance.
By product type, the coronary guidewires subsegment dominated the market in 2024. Coronary guidewires dominate due to their critical role in minimally invasive procedures, the rising prevalence of coronary artery disease, and technological advancements ensuring precision, safety, and improved patient outcomes in cardiovascular interventions.
Based on coating, the coated subsegment dominated the market in 2024. Coated guidewires led the market as surface coatings enhance durability, reduce friction, improve navigation through complex vessels, and minimize complications, making them the preferred choice among physicians for efficiency and patient safety.
In terms of application, the coronary artery disease subsegment dominated the market in 2024. The coronary artery disease segment dominated as increasing global incidence, lifestyle risk factors, and demand for advanced interventional cardiology solutions drove adoption of guidewires and sheaths tailored for coronary procedures.
By end user, the hospitals subsegment dominated the market in 2024. Hospitals dominated the market since they handle the majority of complex cardiovascular interventions, offer advanced infrastructure, skilled specialists, and comprehensive patient care, making them the primary setting for guidewire and sheath usage.
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 723.0 Million |
| Market Size by 2033 | US$ 1,447.8 Million |
| CAGR (2025 - 2033) | 8.2% |
| Historical Data | 2022-2023 |
| Forecast period | 2025-2033 |
| Segments Covered | By By Product Type
|
|
Regions and Countries Covered
| |
| North America | US, Canada, Mexico |
| Market leaders and key company profiles |
|
The "North America Guidewires and Introducer Sheaths Market Size and Forecast (2022–2033)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the North America Guidewires and Introducer Sheaths market report is divided into: The US, Canada, and Mexico. The US held the largest share in 2024.
The US dominates the North American market due to its scale, advanced hospital networks, and leadership in cardiovascular innovation. Hospitals across major metropolitan hubs are equipped with state‑of‑the‑art catheterization labs, driving demand for guidewires and introducer sheaths optimized for efficiency and reproducibility. Procurement is structured, with GPOs and IDNs negotiating contracts that emphasize cost efficiency, clinical evidence, and vendor service commitments. Clinicians in the US emphasize reproducibility and precision, selecting devices that integrate seamlessly into standardized care pathways and support rapid patient recovery. A distinctive feature of the US market is its integration of digital health and quality registries. Devices are tracked against patient outcomes, complication rates, and procedural efficiency, influencing procurement strategies. Growth is reinforced by partnerships with international vendors, academic hospitals piloting new consumables, and simulation‑based training programs that elevate clinician proficiency. Constraints include strict cost‑effectiveness evaluations, competitive pricing pressures, and the need for vendors to comply with rigorous regulatory standards. Nonetheless, the US benefits from a culture of innovation, collaborative procurement, and patient‑centric care models. Vendors that invest in localized training, sustainability initiatives, and evidence‑driven product positioning are best placed to deepen adoption and sustain share in this dominant North American market.
The North America Guidewires and Introducer Sheaths market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the North America guidewires and introducer sheaths market are:
The North America Guidewires and Introducer Sheaths Market is valued at US$ 723.0 Million in 2024, it is projected to reach US$ 1,447.8 Million by 2033.
As per our report North America Guidewires and Introducer Sheaths Market, the market size is valued at US$ 723.0 Million in 2024, projecting it to reach US$ 1,447.8 Million by 2033. This translates to a CAGR of approximately 8.2% during the forecast period.
The North America Guidewires and Introducer Sheaths Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Guidewires and Introducer Sheaths Market report:
The North America Guidewires and Introducer Sheaths Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Guidewires and Introducer Sheaths Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Guidewires and Introducer Sheaths Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)