The North America Cancer Targeted Therapy Market size is expected to reach US$ 97,104.7 million by 2031 from US$ 39,623.9 million in 2024. The market is estimated to record a CAGR of 13.8 % from 2025 to 2031.
The Western European cancer targeted therapy market is high‑value and innovation‑driven, characterized by strong healthcare systems, early adoption of precision medicine, and robust regulatory frameworks within the broader oncology landscape. Countries such as Germany, France, the UK, Italy, Spain, and the Nordics contribute significantly to the regional market, reflecting strong demand for monoclonal antibodies, tyrosine kinase inhibitors (TKIs), and advanced immuno‑oncology combinations.
Targeted therapies are increasingly displacing traditional chemotherapy as standard care across key cancer indications—including lung, breast, colorectal, and hematologic cancers—due to their tumor‑specific mechanisms and improved safety profiles. Public healthcare systems and statutory insurance coverage in most Western European countries support access to high‑cost targeted agents, particularly those with compelling clinical benefits.
Various reforms in regulatory pathways—such as adaptive licensing and joint health technology assessments—have reduced barriers to market entry for new targeted agents, further accelerating uptake. Western Europe continues to evolve as a strategic market for global pharmaceutical companies seeking early adoption, real‑world evidence generation, and strong commercial returns in precision oncology.
Key segments that contributed to the derivation of the Western Europe Cancer Targeted Therapy market analysis are therapy type, indication, and distribution channel.
Advancements in genomic profiling and biomarker discovery are key market drivers. Widespread adoption of next‑generation sequencing (NGS), liquid biopsy assays, and comprehensive molecular diagnostics permits clinicians to identify actionable genetic mutations such as EGFR, ALK, HER2, PIK3CA, and NTRK fusions, enabling precision therapy tailored to tumor biology. These technologies support patient stratification for targeted agents, improving clinical outcomes and reducing unnecessary exposure to less effective treatments.
Healthcare systems in Western Europe—particularly in Germany, France, and the UK—have integrated genomic profiling into clinical pathways for common cancers such as lung and breast cancer. Companion diagnostic programs are supported by public reimbursement, ensuring that patients eligible for targeted therapies receive appropriate molecular testing. This integration accelerates the transition from one‑size‑fits‑all chemotherapy to precision oncology, elevating the role of targeted therapies in standard care.
Liquid biopsies—which analyze circulating tumor DNA—have expanded monitoring capabilities beyond initial diagnosis to include treatment response and resistance detection. The accumulation of region‑specific genomic data also fuels research collaboration, enabling novel target discovery and optimizing clinical trial design.
Investments in digital health platforms and AI‑driven analytics further enhance biomarker discovery efficiency, making it easier to translate discoveries into clinical practice. Collectively, these developments reinforce the clinical value of targeted therapies, establish strong physician confidence, and drive sustained market growth.
Expanding targeted therapies into rare cancers and tumors defined by specific genetic mutations represents a strategic market opportunity. Historically, rare cancers have been underserved due to limited patient numbers and high development costs. However, advanced genomic profiling has revealed that many rare malignancies share actionable mutations found in more common tumors, such as NTRK fusions, RET rearrangements, and BRAF alterations. Tumor‑agnostic therapies that target these genetic drivers can be used across multiple rare cancer types, significantly expanding the addressable patient population.
Regulatory incentives such as orphan drug designations and accelerated approval pathways in the European Union encourage pharmaceutical investment in rare cancer therapeutics. These incentives and approvals have resulted in an expanding pipeline of precision oncology agents evaluated in innovative clinical trial designs such as basket and umbrella trials, which allow simultaneous testing of therapies across diverse tumor types sharing a genetic signature.
Western European healthcare systems and cancer centers are well-positioned to adopt these advanced therapies due to their access to molecular diagnostics and strong clinical research networks. Early access programs and real‑world evidence generation support regulatory decision‑making and reimbursement negotiations, enhancing market entry strategies for rare cancer therapies.
Furthermore, patient advocacy organizations and clinician education programs raise awareness of rare cancer diagnostics and treatment options, increasing demand for targeted agents. As genomics becomes integral to cancer care, therapies for rare, mutation‑defined cancers will play a growing role in regional treatment paradigms, fostering deeper market penetration and stronger revenue growth for targeted therapy portfolios.
The Western Europe Cancer Targeted Therapy market demonstrates steady growth, with size and share analysis highlighting evolving trends and competitive dynamics among key players. The report examines subsegments categorized within therapy type, indication, and distribution channel, offering insights into their contribution to overall market performance.
By therapy type, the monoclonal antibodies subsegment dominated the market in 2024, driven by their clinical versatility and strong evidence base across multiple indications, including breast, colorectal, and lung cancers.
Based on indication, the lung cancer subsegment dominated the market in 2024, driven by the high prevalence and the availability of numerous targeted agents addressing key genetic drivers (EGFR, ALK, ROS1, PD‑L1).
By distribution channel, the hospital pharmacies subsegment dominated the market in 2024, driven by targeted therapies, especially monoclonal antibodies and ADCs, which require clinical administration, monitoring, and adherence support.
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 21,460.8 Million |
| Market Size by 2031 | US$ 50,427.8 Million |
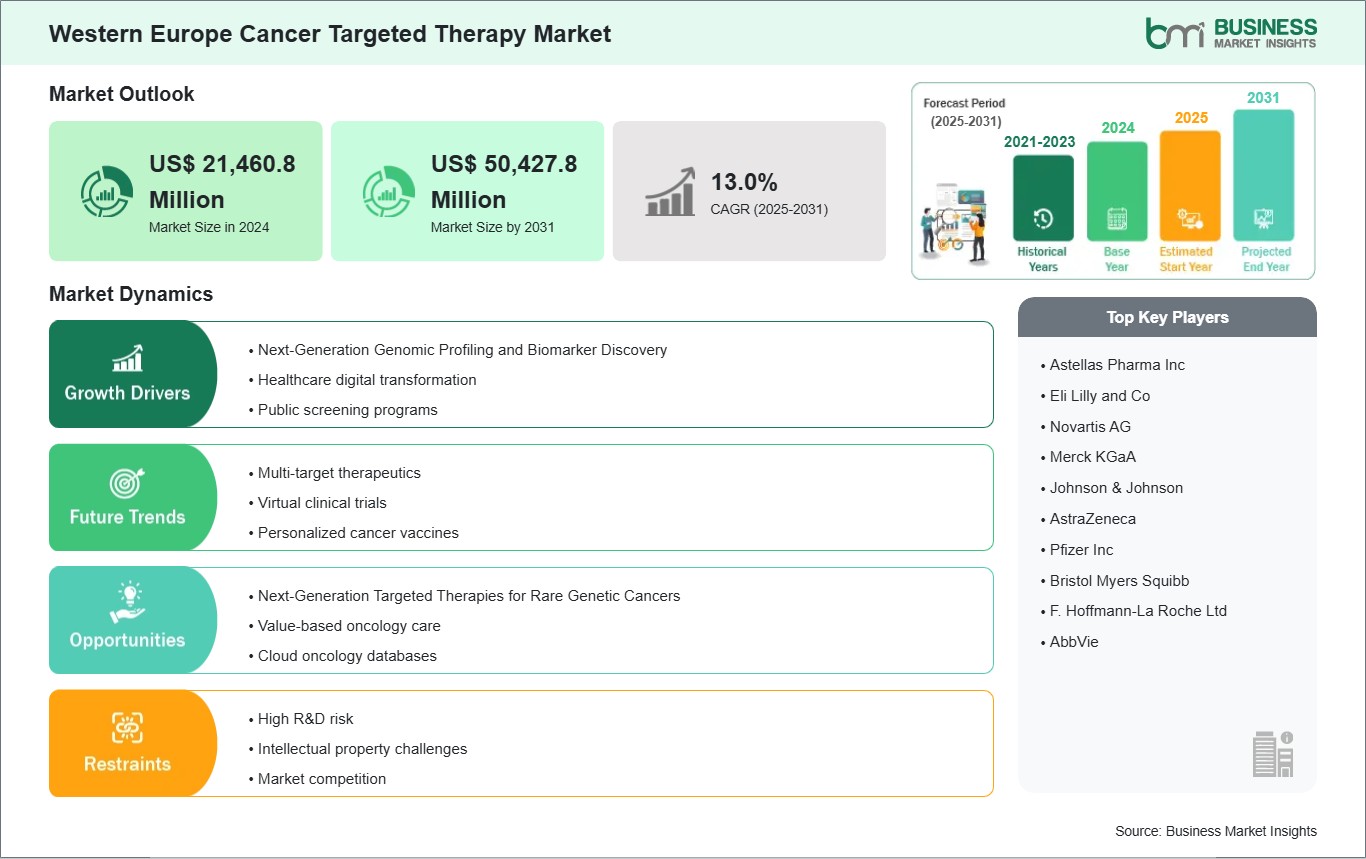
| CAGR (2025 - 2031) | 13.0% |
| Historical Data | 2021-2023 |
| Forecast period | 2025-2031 |
| Segments Covered | By Therapy Type
|
|
Regions and Countries Covered
| |
| Western Europe | Belgium, the Netherlands, Luxembourg, Germany, France, Italy, Spain, Switzerland, Sweden, Austria, the United Kingdom, Denmark, Portugal, Norway, Finland |
| Market leaders and key company profiles |
|
The "Western Europe Cancer Targeted Therapy Market Size and Forecast (2021–2031)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the Western Europe Cancer Targeted Therapy market report is divided into: Belgium, the Netherlands, Luxembourg, Germany, France, Italy, Spain, Switzerland, Sweden, Austria, the UK, Denmark, Portugal, Norway, and Finland. Germany held the largest share in 2024.
Germany continues to lead the market due to its large population, high healthcare spending, and dense network of cancer centers and clinical research institutions. German hospitals and oncology centers routinely implement molecular profiling and precision treatment strategies, supported by favorable reimbursement frameworks and early benefit assessments that accelerate access to breakthrough therapies.
France is another major contributor, with a national genomic screening rollout and streamlined health technology assessment (HTA) processes that shorten time to market for targeted therapies. Centralized purchasing and coordinated oncology networks facilitate widespread distribution across urban and regional hospitals.
In the UK, despite post‑Brexit regulatory alignment shifts, mutual recognition arrangements with EMA dossiers ensure continued access to innovative targeted oncology drugs. The National Health Service (NHS) and bodies such as NICE influence adoption through cost‑effectiveness evaluations and reimbursement guidance.
Italy and Spain show increasing targeted therapy uptake supported by enhanced oncology spending and growing biosimilar use that improves affordability. Healthcare reforms emphasize precision medicine and early adoption of validated targeted agents, particularly for breast and lung cancers.
Nordic and Benelux countries leverage digital registries and coordinated screening initiatives to optimize patient selection and therapy outcomes. Across Western Europe, hospital pharmacies remain the dominant distribution channel for intravenous biologics, while oral-targeted therapies are increasingly accessible through retail and specialty pharmacies. Robust public insurance systems and multinationals' regional subsidiaries ensure broad access, making Western Europe a mature and growth‑oriented market for cancer-targeted therapies.

The Western Europe Cancer Targeted Therapy market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the Western Europe cancer targeted therapy market are:
The Western Europe Cancer Targeted Therapy Market is valued at US$ 21,460.8 Million in 2024, it is projected to reach US$ 50,427.8 Million by 2031.
As per our report Western Europe Cancer Targeted Therapy Market, the market size is valued at US$ 21,460.8 Million in 2024, projecting it to reach US$ 50,427.8 Million by 2031. This translates to a CAGR of approximately 13.0% during the forecast period.
The Western Europe Cancer Targeted Therapy Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Western Europe Cancer Targeted Therapy Market report:
The Western Europe Cancer Targeted Therapy Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Western Europe Cancer Targeted Therapy Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Western Europe Cancer Targeted Therapy Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)