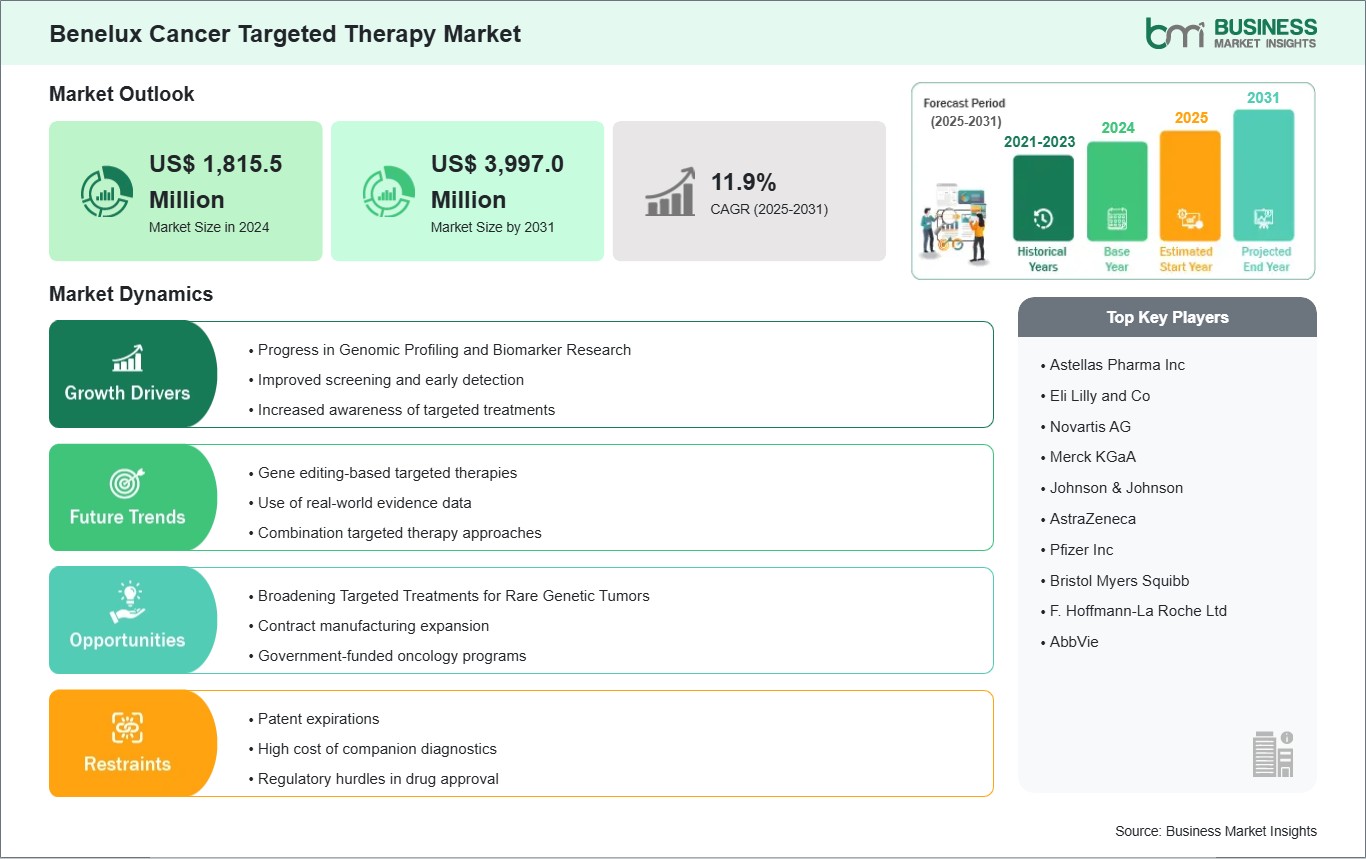
The Benelux Cancer Targeted Therapy Market size is expected to reach US$ 3,997.0 million by 2031 from US$ 1,815.5 million in 2024. The market is estimated to record a CAGR of 11.9% from 2025 to 2031.
The Benelux cancer targeted therapy market has experienced significant growth due to rapid advancements in precision oncology, genomic profiling, and biomarker-guided therapeutics. Belgium, the Netherlands, and Luxembourg benefit from highly developed healthcare systems, supportive regulatory frameworks, and strong research collaborations, which facilitate rapid adoption of monoclonal antibodies, tyrosine kinase inhibitors, and immune checkpoint inhibitors. Hospitals and specialized oncology centers dominate therapy delivery, whereas private clinics and diagnostic labs increasingly contribute through genomic testing and companion diagnostics.
Government support through national cancer plans, reimbursement frameworks, and precision medicine initiatives has accelerated access to advanced targeted therapies. Moreover, real-world evidence generation from multicenter clinical trials strengthens confidence in therapy outcomes and drives adoption. The market is further supported by active collaboration between pharmaceutical companies, biotech firms, and academic institutions, focusing on biomarker discovery, tumor-agnostic therapies, and pipeline expansion for rare cancers.
The region's research infrastructure allows early adoption of tumor-specific therapies for lung, breast, colorectal, and hematological cancers. Regulatory alignment with EMA protocols ensures rapid approvals, particularly for therapies addressing rare genetic mutations. The combination of infrastructure readiness, regulatory support, and collaborative research initiatives positions the Benelux region as a strategically important market within Europe for cancer-targeted therapies.
Key segments that contributed to the derivation of the Benelux Cancer Targeted Therapy market analysis are therapy type, indication, and distribution channel.
Widespread adoption of next-generation sequencing (NGS), liquid biopsies, whole-exome sequencing, and other molecular diagnostics enables oncologists to identify actionable genetic mutations—including EGFR, ALK, RET, BRAF, FGFR, and PIK3CA—which influence tumor behavior and treatment response. This shift toward precision medicine allows clinicians to select therapies tailored to the patient's tumor genomics, increasing efficacy while minimizing systemic toxicity.
Biomarker-driven stratification improves clinical trial efficiency, reduces development risks, and enhances regulatory success. Companion diagnostics are increasingly required for approval, reinforcing the commercial ecosystem for precision oncology. Liquid biopsy technologies allow real-time disease monitoring, minimal residual disease detection, and identification of resistance mechanisms, extending the utility of targeted therapies and improving patient outcomes.
Additionally, AI-driven analytics and real-world genomic databases accelerate biomarker validation and novel target discovery across rare and difficult-to-treat cancers. Reimbursement for genomic testing and precision diagnostics raises adoption rates, expanding the pool of eligible patients. The integration of these technological and diagnostic advancements ensures that the region remains a leader in precision oncology, supporting the sustained adoption and innovation of targeted therapies across common and rare cancer indications.
Rare cancers have traditionally faced limited therapeutic options due to small patient populations and high development costs. However, genomic profiling has revealed that several rare tumors share actionable mutations, enabling tumor-agnostic, mutation-driven treatment strategies.
Tumor-agnostic therapies targeting biomarkers such as NTRK fusions, RET rearrangements, BRAF mutations, and FGFR alterations have gained approvals for multiple malignancies, opening opportunities for single therapies to treat diverse rare cancer types. Supportive regulatory frameworks—including orphan drug designation, accelerated approval pathways, and extended market exclusivity—enhance the commercial feasibility of rare cancer therapy development.
Clinical outcomes for targeted therapies in genetically driven tumors often surpass those of conventional chemotherapy, offering improved efficacy and tolerability. Comprehensive molecular testing and widespread NGS adoption have increased the detection of rare mutations, expanding the eligible patient population. Innovative trial designs such as basket and umbrella trials reduce costs and shorten timelines for rare indications.
These advances allow pharmaceutical companies to extend indications across multiple low-incidence cancers, generating new revenue streams, strengthening pipelines, and enhancing long-term sustainability in the market. As Benelux integrates precision oncology into standard treatment guidelines, the market is well-positioned to capitalize on these opportunities, particularly for tumor-agnostic therapies and personalized cancer treatment strategies.
The Benelux Cancer Targeted Therapy market demonstrates steady growth, with size and share analysis highlighting evolving trends and competitive dynamics among key players. The report examines subsegments categorized within therapy type, indication, and distribution channel, offering insights into their contribution to overall market performance.
By therapy type, the monoclonal antibodies subsegment dominated the market in 2024, driven by their high clinical efficacy in multiple cancer indications and increasing adoption of immuno-oncology strategies.
Based on indication, the lung cancer subsegment dominated the market in 2024, driven by the high disease prevalence, environmental risk factors, and the availability of targeted therapies addressing key genetic mutations.
By distribution channel, the hospital pharmacies subsegment dominated the market in 2024, driven by the specialized oncology infrastructure, treatment initiation, and continuous patient monitoring, which are essential for complex targeted therapies.
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 1,815.5 Million |
| Market Size by 2031 | US$ 3,997.0 Million |
| CAGR (2025 - 2031) | 11.9% |
| Historical Data | 2021-2023 |
| Forecast period | 2025-2031 |
| Segments Covered | By Therapy Type
|
|
Regions and Countries Covered
| |
| Benelux | Belgium, Netherlands, Luxembourg |
| Market leaders and key company profiles |
|
The "Benelux Cancer Targeted Therapy Market Size and Forecast (2021–2031)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the Benelux Cancer Targeted Therapy market report is divided into: Belgium, the Netherlands, and Luxembourg. The Netherlands held the largest share in 2024.
Benelux comprises Belgium, the Netherlands, and Luxembourg, each contributing unique dynamics to the cancer targeted therapy market. Belgium leads in precision oncology adoption, driven by strong national cancer programs, molecular tumor boards, and reimbursement frameworks that facilitate early access to monoclonal antibodies and immune checkpoint inhibitors. Specialized hospitals and oncology centers dominate therapy delivery, supported by growing adoption of NGS and companion diagnostics.
The Netherlands emphasizes research-driven adoption, integrating multicenter clinical trials, real-world evidence generation, and tumor-agnostic therapy programs. Hospitals, private clinics, and diagnostic laboratories collaborate to accelerate biomarker identification, supporting common and rare cancer indications. Luxembourg—although smaller in population—benefits from high healthcare expenditure, cross-border clinical trial integration, and centralized treatment infrastructure, ensuring access to advanced therapies.
Regulatory alignment with EMA protocols and favorable reimbursement policies allows rapid approvals for both common and rare genetically driven cancers. Collaborative research initiatives between academic institutions, regional biotech companies, and multinational pharma enhance biomarker discovery, pipeline expansion, and therapy personalization.
Hospitals remain the primary end users, while private diagnostic centers and research hospitals are increasingly adopting targeted therapies, improving patient access and treatment outcomes. The combined effect of robust healthcare infrastructure, supportive regulations, precision medicine adoption, and collaborative research ecosystems positions the Benelux region as a strategically important region for targeted cancer therapy innovation in Europe. Differentiated strategies—premium adoption in Belgium, research-led programs in the Netherlands, and cross-border integration in Luxembourg—ensure comprehensive market coverage and long-term growth.

The Benelux Cancer Targeted Therapy market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the Benelux cancer targeted therapy market are:
The Benelux Cancer Targeted Therapy Market is valued at US$ 1,815.5 Million in 2024, it is projected to reach US$ 3,997.0 Million by 2031.
As per our report Benelux Cancer Targeted Therapy Market, the market size is valued at US$ 1,815.5 Million in 2024, projecting it to reach US$ 3,997.0 Million by 2031. This translates to a CAGR of approximately 11.9% during the forecast period.
The Benelux Cancer Targeted Therapy Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Benelux Cancer Targeted Therapy Market report:
The Benelux Cancer Targeted Therapy Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Benelux Cancer Targeted Therapy Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Benelux Cancer Targeted Therapy Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)