The Eastern Europe intravenous catheters market size is expected to reach US$ 1,650.5 million by 2033 from US$ 852.7 million in 2024. The market is estimated to record a CAGR of 7.6% from 2025 to 2033.
The intravenous catheters market in Eastern Europe is experiencing significant growth driven by the increase in hospitalization and surgical procedures; especially in ICUs and oncology, higher adoption of peripherally inserted central catheters (PICCs) and midline catheters, and rising focus on infection control driving demand for coated/antimicrobial catheters. Eastern Europe’s intravenous catheter market reflects heterogeneous systems transitioning from legacy practices to modern, outcome-oriented care. Peripheral catheters constitute the foundational device category, supporting high-volume medical wards, surgical units, and emergency departments where swift cannulation and short dwell dominate. Coated catheters are gaining share in urban tertiary hospitals engaged in accreditation cycles and quality incentive schemes; antimicrobial and hydrophilic surfaces are adopted to curb phlebitis and catheter-related bloodstream infections that strain limited ICU capacity. Multi-lumen devices see targeted deployment in oncology hubs and advanced ICUs, aligning with centralization of complex care in capital cities and regional university hospitals. The end-user base is overwhelmingly public, with clinics attached to hospital networks and polyclinics expanding infusion capacity. Procurement is increasingly professionalized: national tenders and hospital consortia emphasize lifecycle cost, training inclusions, and documented performance under local conditions. Distributors remain pivotal, bridging language, regulatory particulars, and last-mile logistics across fragmented geographies. Market access is sensitive to formularies and coding; vendors that tailor submission dossiers, participate in pilot studies, and offer clinician education gain influence in guideline updates. Affordability and reliability trump novelty in many districts, but visible wins on infection rates and nurse workload accelerate coated adoption where budgets allow. Regional variance is marked—EU members align faster with MDR expectations and quality metrics, while non-EU neighbours progress through phased upgrades. Overall, the region values pragmatic devices that reduce complications, sustain throughput, and fit constrained staffing models, rewarding portfolios that combine robust peripheral lines, selectively deployed coatings, and multi-lumen options for consolidated specialty care.
Eastern Europe Intravenous Catheters Market Strategic Insights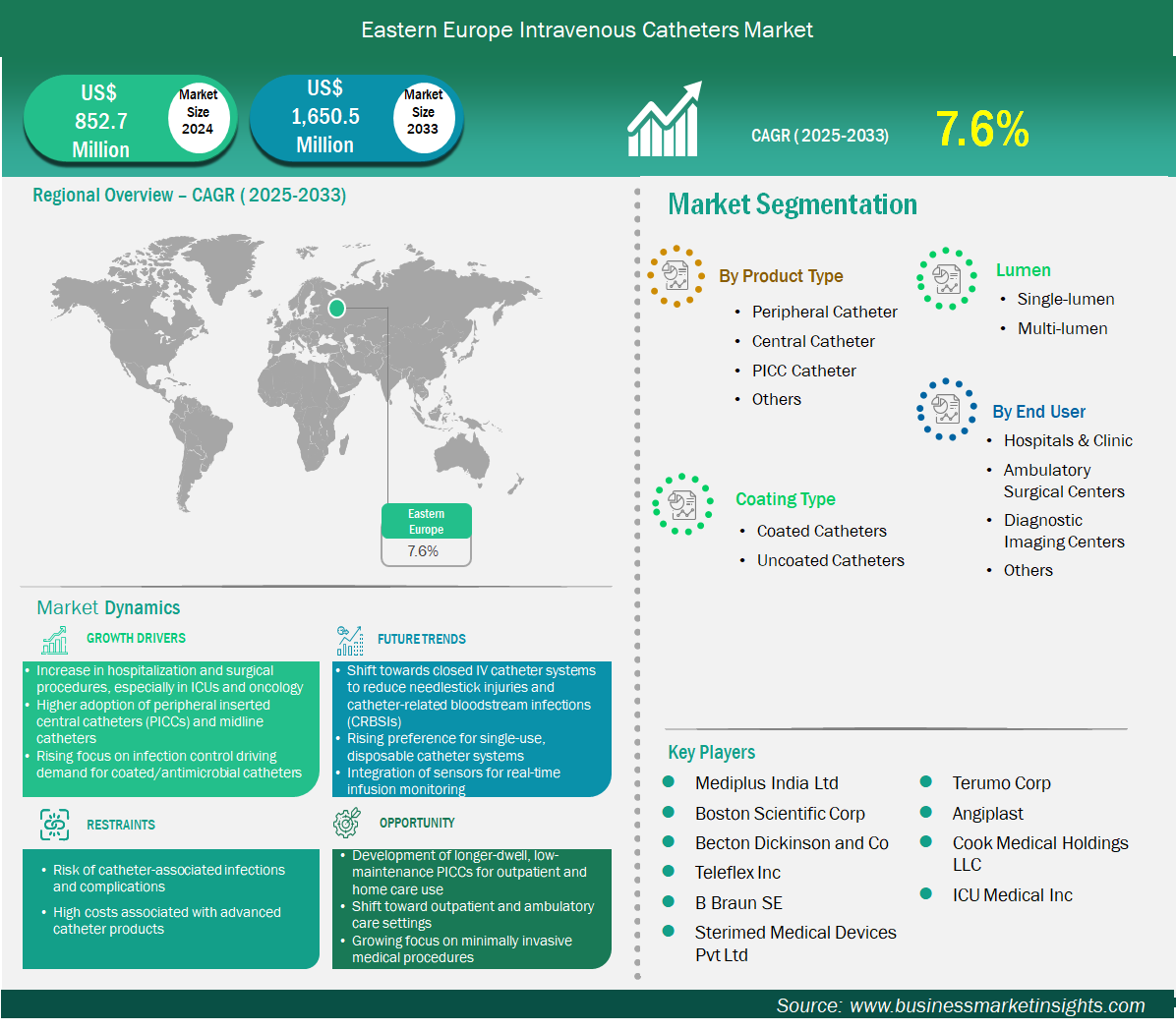
Key segments that contributed to the derivation of the intravenous catheters market analysis are product, coating type, lumen, and end user.
The Eastern Europe intravenous catheters market is segmented into Russia, Poland, Ukraine, Romania, the Czech Republic, Slovakia, Bulgaria, and Greece. Eastern Europe’s trajectory will be steered by EU cohesion funding, hospital consolidation, and workforce optimization. Peripheral catheter demand will remain resilient as hospitals tackle bed flow pressures and shrink average length of stay, favouring easy-insertion, vein-friendly designs that reduce failure and repeat cannulation. Coated catheters will expand with MDR-aligned procurement and infection control mandates—expect bundled tenders that link price to verified reductions in CRBSI and phlebitis, plus vendor-supported audits. Multi-lumen use will rise in oncology and critical care as treatment protocols intensify and referral pathways concentrate complex cases; devices with dependable lumen separation and simplified maintenance will gain preference. Supply chain resilience is becoming a board-level topic—institutions will favour regional warehousing, guaranteed replenishment windows, and dual-distributor frameworks to mitigate transport and customs delays. Training remains a growth lever: simulation-driven insertion courses and standardized securement practices can unlock coated adoption and reduce complication-related bed days. Digital initiatives—barcode tracking, EMR integration of line data, and adverse event dashboards—will spread from capitals to tier-2 cities, enabling outcome-based purchasing. Price sensitivity endures; vendors must articulate total cost advantages through fewer complications, lower reinsertions, and reduced nurse time. Sustainability criteria will enter tender scoring in EU members, encouraging lighter packaging and recyclability. Strategic success depends on blending affordability with measurable clinical gains, local service presence, and policy fluency—aligning bids with national cancer plans, infection targets, and workforce strategies. Expect steady growth, rooted in modernization projects and quality-driven procurement that elevates coated and multi-lumen segments without displacing peripheral mainstays.
Based on region, the Eastern Europe intravenous catheters market is further segmented into Russia, Poland, Ukraine, Romania, the Czech Republic, Slovakia, Bulgaria, and Greece. Russia held the largest share in 2024.
Russia’s large public hospital networks and oncology centers drive structured demand: peripheral lines dominate ward-level use, while coated devices gain traction in teaching hospitals pursuing accreditation benchmarks. Romania and Bulgaria prioritize affordability but are piloting coated catheters in capitals to address infection indicators; university hospitals anchor multi-lumen adoption for complex regimens. Czechia and Slovakia showcase tighter MDR alignment and mature procurement committees, valuing vendor education packages and guaranteed supply continuity. Across the region, infusion clinics attached to hospital systems are expanding, but independent private centers remain limited outside major cities. Tender success hinges on local references, clinician champions, and post-award training fidelity. Value narratives that emphasize fewer complications, consistent dwell times, and ergonomic insertion resonate with nurse leaders tasked with throughput under staffing constraints. Policy touchpoints—cancer plans, MDR timelines, and infection reporting—shape portfolio choices; vendors that align with national priorities and provide audit-ready data gain durable contracts. Country diversity necessitates tiered offerings: baseline peripheral ranges for volume wards, coated options for tertiary hubs, and multi-lumen SKUs for oncology and ICU anchors.
Eastern Europe Intravenous Catheters Market Report Highlights| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 852.7 Million |
| Market Size by 2033 | US$ 1,650.5 Million |
| CAGR (2025 - 2033) | 7.6% |
| Historical Data | 2022-2023 |
| Forecast period | 2025-2033 |
| Segments Covered |
By Product
|
|
Regions and Countries Covered
|
|
| Eastern Europe | Russia, Poland, Ukraine, Romania, Czech Republic, Slovakia, Bulgaria, Greece |
| Market leaders and key company profiles |
|
Mediplus India Ltd, Boston Scientific Corp, Becton Dickinson and Co, Teleflex Inc, B Braun SE, Sterimed Medical Devices Pvt Ltd, Terumo Corp, Angiplast, Cook Medical Holdings LLC, ICU Medical Inc, are among the key players operating in the market. These players adopt strategies such as expansion, product innovation, and mergers and acquisitions to stay competitive in the market and offer innovative products to their consumers.
The following methodology has been followed for the collection and analysis of data presented in this report:
The research process begins with comprehensive secondary research, utilizing both internal and external sources to gather qualitative and quantitative data for each market. Commonly referenced secondary research sources include, but are not limited to:
Note: All financial data included in the Company Profiles section has been standardized to US$. For companies reporting in other currencies, figures have been converted to US$ using the relevant exchange rates for the corresponding year.
Business Market Insights conducts a significant number of primary interviews each year with industry stakeholders and experts to validate and analyze the data and gain valuable insights. These research interviews are designed to:
Primary research is conducted via email interactions and telephone interviews with industry experts across various markets, categories, segments, and sub-segments in different regions. Participants typically include:
The Eastern Europe Intravenous Catheters Market is valued at US$ 852.7 Million in 2024, it is projected to reach US$ 1,650.5 Million by 2033.
As per our report Eastern Europe Intravenous Catheters Market, the market size is valued at US$ 852.7 Million in 2024, projecting it to reach US$ 1,650.5 Million by 2033. This translates to a CAGR of approximately 7.6% during the forecast period.
The Eastern Europe Intravenous Catheters Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Eastern Europe Intravenous Catheters Market report:
The Eastern Europe Intravenous Catheters Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Eastern Europe Intravenous Catheters Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Eastern Europe Intravenous Catheters Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)