The Western Europe interventional cardiology devices market size is expected to reach US$ 7,968.3 million by 2033 from US$ 4,118.9 million in 2024. The market is estimated to record a CAGR of 7.6% from 2025 to 2033.
Executive Summary and Western Europe Interventional Cardiology Devices Market Analysis:
Western Europe remains one of the strongest and most mature subregions in the interventional cardiology devices market. Countries such as Germany, France, the United Kingdom, Italy, and Spain lead in procedural volumes, clinical adoption of advanced technologies, and healthcare infrastructure.
The presence of specialized cardiac centers, experienced interventional cardiologists, and comprehensive reimbursement systems support widespread use of stents, diagnostic imaging tools, and transcatheter heart valves. Ongoing innovation, government health programs, and aging populations drive sustained demand. Despite pricing pressures and market saturation in some areas, Western Europe continues to be a core contributor to global revenues and a center for clinical research and device adoption.
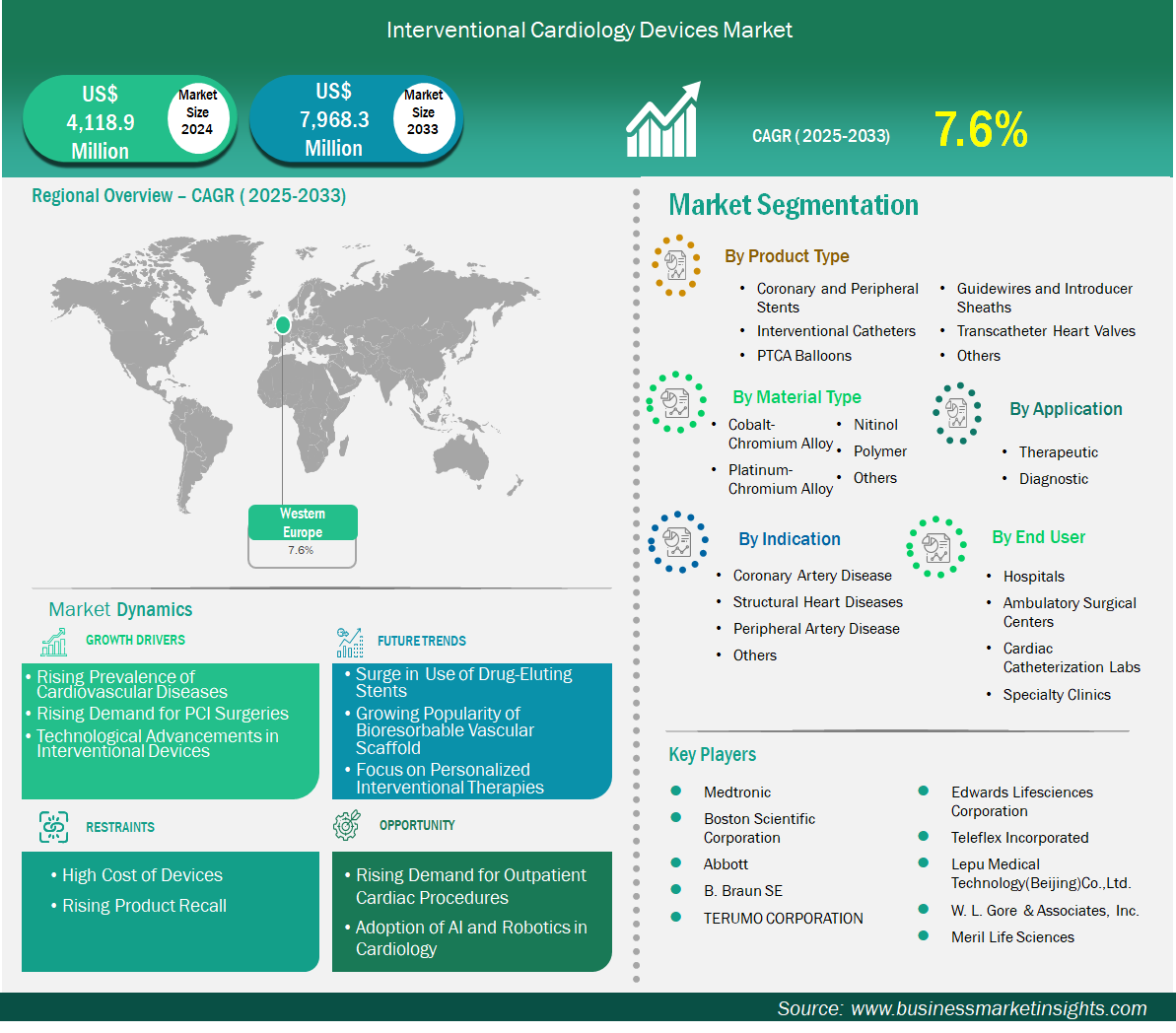
Key segments that contributed to the derivation of the interventional cardiology devices market analysis are product type, material type, application, indication, and end user.
Western Europe Interventional Cardiology Devices Market Outlook
The Western European interventional cardiology devices market is a mature and highly developed sector, led by countries with advanced healthcare systems like Germany, France, and the UK. The market is characterized by the widespread adoption of innovative, high-value devices and a strong emphasis on evidence-based medicine. This is a region with a substantial elderly population and a high prevalence of cardiovascular diseases, which naturally creates a significant demand for interventional procedures. The market is propelled by continuous technological advancements and the development of next-generation devices, such as drug-eluting balloons and bioresorbable stents, which improve patient outcomes and address complex clinical challenges. Favorable reimbursement policies across many Western European countries for these advanced and often premium-priced devices also play a crucial role, ensuring that a broad patient base has access to the latest therapies.
Western Europe Interventional Cardiology Devices Market Country Insights
Based on country, the Western Europe interventional cardiology devices market is segmented into Belgium, the Netherlands, Luxembourg, Germany, France, Italy, Spain, Switzerland, Sweden, Austria, United Kingdom, Denmark, Portugal, Norway, and Finland. Germany held the largest share in 2024.
Germany dominates the Western Europe market in the interventional cardiology device landscape, characterized by a sophisticated healthcare system, a high volume of cardiac procedures, and a strong adoption rate of advanced medical technologies. The country's robust economy and comprehensive health insurance coverage further contribute to its leading position.
Cardiovascular diseases remain the leading cause of death in Germany, accounting for approximately 40% of all fatalities. This significant disease burden drives a continuous demand for diagnostic and interventional solutions. In 2022, the total number of interventional cardiology procedures performed in Germany was over 1.47 million. Germany also ranked high in Europe for transluminal coronary angioplasty procedures, with 379.3 procedures per 100,000 inhabitants in 2022, indicating a strong reliance on PCI for CAD treatment. This blend of high disease prevalence, advanced medical infrastructure, and a proactive approach to adopting cutting-edge interventional techniques solidifies Germany's role as a driving force in the global interventional cardiology devices market.
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 4,118.9 Million |
| Market Size by 2033 | US$ 7,968.3 Million |
| CAGR (2025 - 2033) | 7.6% |
| Historical Data | 2022-2023 |
| Forecast period | 2025-2033 |
| Segments Covered |
By Product Type
|
|
Regions and Countries Covered
|
|
| Western Europe | Austria, Belgium, Denmark, Finland, France, Germany, Italy, Luxembourg, Netherlands, Norway, Portugal, Spain, Sweden, Switzerland, United Kingdom |
| Market leaders and key company profiles |
|
Medtronic, Boston Scientific Corporation, Abbott, B. Braun SE, TERUMO CORPORATION, Edwards Lifesciences Corporation, Teleflex Incorporated, Lepu Medical Technology (Beijing) Co., Ltd., W. L. Gore & Associates, Inc., and Meril Life Sciences are among the key players operating in the market. These players adopt strategies such as expansion, product innovation, and mergers and acquisitions to stay competitive in the market and offer innovative products to their consumers.
Western Europe Interventional Cardiology Devices Market Research Methodology:
The following methodology has been followed for the collection and analysis of data presented in this report:
The research process begins with comprehensive secondary research, utilizing both internal and external sources to gather qualitative and quantitative data for each market. Commonly referenced secondary research sources include, but are not limited to:
Note: All financial data included in the Company Profiles section has been standardized to US$. For companies reporting in other currencies, figures have been converted to US$ using the relevant exchange rates for the corresponding year.
Business Market Insights conducts a significant number of primary interviews each year with industry stakeholders and experts to validate and analyze the data and gain valuable insights. These research interviews are designed to:
Primary research is conducted via email interactions and telephone interviews with industry experts across various markets, categories, segments, and sub-segments in different regions. Participants typically include:
The Western Europe Interventional Cardiology Devices Market is valued at US$ 4,118.9 Million in 2024, it is projected to reach US$ 7,968.3 Million by 2033.
As per our report Western Europe Interventional Cardiology Devices Market, the market size is valued at US$ 4,118.9 Million in 2024, projecting it to reach US$ 7,968.3 Million by 2033. This translates to a CAGR of approximately 7.6% during the forecast period.
The Western Europe Interventional Cardiology Devices Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Western Europe Interventional Cardiology Devices Market report:
The Western Europe Interventional Cardiology Devices Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Western Europe Interventional Cardiology Devices Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Western Europe Interventional Cardiology Devices Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)