The Western Europe pulmonary pressure monitors market size is expected to reach US$ 825.2 million by 2033 from US$ 493.8 million in 2024. The market is estimated to record a CAGR of 5.9% from 2025 to 2033.
The pulmonary pressure monitors market in Western Europe is experiencing significant growth driven by the growing incidence of pulmonary hypertension and COPD, advancements in non-invasive and portable monitoring technologies, and rising demand for remote respiratory monitoring in home care. The pulmonary pressure monitors market in Western Europe is influenced by a mature healthcare ecosystem with high clinical standards and a strong demand for precision diagnostics. Germany, France, the UK, Italy, and Spain are the leading countries in cardiopulmonary care in the region, supported by well-funded public health systems and advanced tertiary care infrastructure. The market is somewhat consolidated, with the top global MedTech companies having direct operations and strategic partnerships with university hospitals and research institutions. Procurement models differ from one country to another, with centralized purchasing in France and Italy, while Germany and the UK are more inclined towards decentralized, hospital-led acquisition. Regulatory compliance under EU MDR guarantees product quality and safety but also adds to the cost and complexity of market entry. Although there is saturation in some segments, the market is still moving forward through innovation, clinical research, and integration of monitoring technologies into broader care pathways.
Western Europe Pulmonary Pressure Monitors Market Strategic Insights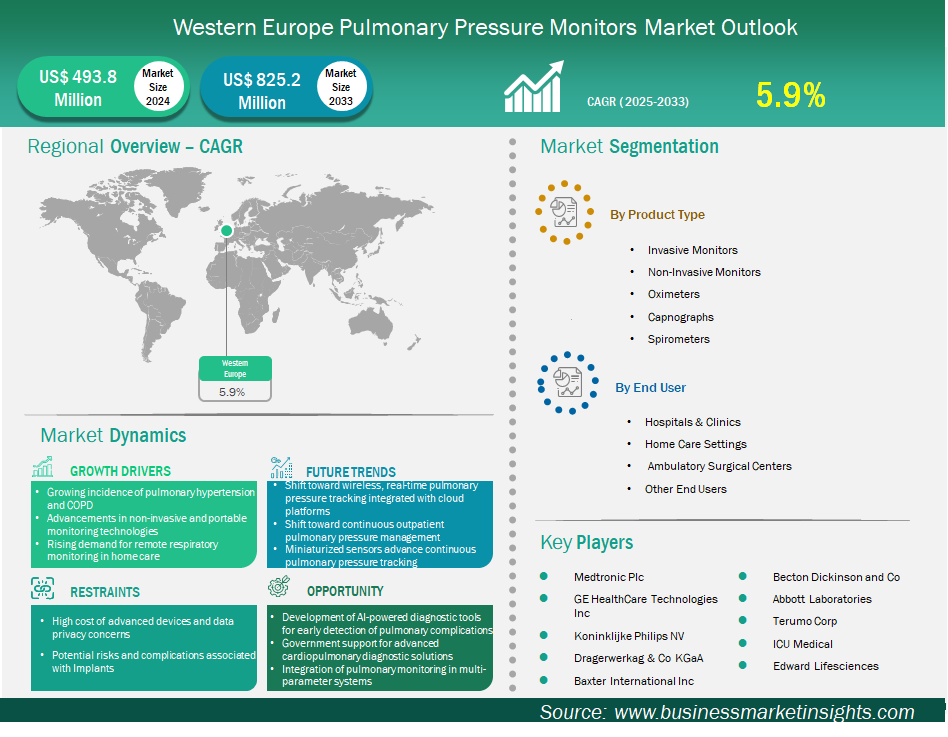
Key segments that contributed to the derivation of the pulmonary pressure monitors market analysis are product and end user.
The digital transformation, the aging demographic, and the shift to integrated care models influence Western Europe's pulmonary pressure monitors market. To improve chronic disease management and reduce hospital readmissions, governments are investing in smart hospital infrastructure and remote monitoring capabilities. Telehealth adoption is at the forefront in the UK and Germany; thus, there is a rising need for connected monitoring devices that facilitate real-time data exchange and patient engagement. France and Italy are working on expanding their reimbursement frameworks to cover advanced diagnostics, while Spain is concentrating on regional health equity through digital outreach programs. With sustainability turning into a procurement priority, manufacturers are being prompted to come up with devices that can be reused and are energy-efficient. There are EU-wide initiatives to support cross-border healthcare and data interoperability, which, in turn, are expected to speed up the adoption of new technologies.
Based on country, the Western Europe pulmonary pressure monitors market is segmented into Belgium, the Netherlands, Luxembourg, Germany, France, Italy, Spain, Switzerland, Sweden, Austria, the United Kingdom, Denmark, Portugal, Norway, and Finland. Germany held the largest share in 2024.
Country-level dynamics across Western Europe have been showing the different priorities and the adoption patterns in all these countries. For instance, Germany is known for its healthcare system, which is not centralized, and a strong private sector that has been responsible for the quick adoption of advanced monitoring technologies. France highly relies on centralized planning and public investment. Hospital universities are the ones playing a major role in clinical validation and procurement. On the other hand, the UK is more concerned with digital health and remote care. This is being done through the integration of pulmonary pressure monitors into national telehealth platforms. Italy is upgrading its healthcare facilities through projects funded by the EU.
Meanwhile, Spain is giving priority to the deprived areas by providing mobile health units and regional diagnostic hubs. Whereas Austria and the Netherlands, as smaller markets, are keeping up with a high level of clinical standards and are among the first to implement interoperable monitoring systems. The reimbursement policies are different and have an effect on market penetration and pricing strategies. The use of the device is mostly in urban centers, while the rural areas are slowly being equipped through outreach programs and digital health pilots. It will be necessary to have country-specific tailored go-to-market strategies that are in line with the regulatory framework, procurement model, and clinical capacity of each country in Western Europe.
Western Europe Pulmonary Pressure Monitors Market Report Highlights| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 493.8 Million |
| Market Size by 2033 | US$ 825.2 Million |
| CAGR (2025 - 2033) | 5.9% |
| Historical Data | 2022-2023 |
| Forecast period | 2025-2033 |
| Segments Covered |
By Product
|
|
Regions and Countries Covered
|
|
| Western Europe | Belgium, Netherlands, Luxembourg, Germany, France, Italy, Spain, Switzerland, Sweden, Austria, United Kingdom, Denmark, Portugal, Norway, Finland |
| Market leaders and key company profiles |
|
Medtronic Plc, GE HealthCare Technologies Inc, Koninklijke Philips NV, Baxter International Inc, Becton Dickinson and Co, Abbott Laboratories, Terumo Corp, Dragerwerk AG & Co KGaA, ICU Medical, and Edwards Lifesciences are among the key players operating in the market. These players adopt strategies such as expansion, product innovation, and mergers and acquisitions to stay competitive in the market and offer innovative products to their consumers.
The following methodology has been followed for the collection and analysis of data presented in this report:
The research process begins with comprehensive secondary research, utilizing both internal and external sources to gather qualitative and quantitative data for each market. Commonly referenced secondary research sources include, but are not limited to:
Note: All financial data included in the Company Profiles section has been standardized to US$. For companies reporting in other currencies, figures have been converted to US$ using the relevant exchange rates for the corresponding year.
Business Market Insights conducts a significant number of primary interviews each year with industry stakeholders and experts to validate and analyze the data and gain valuable insights. These research interviews are designed to:
Primary research is conducted via email interactions and telephone interviews with industry experts across various markets, categories, segments, and sub-segments in different regions. Participants typically include:
The Western Europe Pulmonary Pressure Monitors Market is valued at US$ 493.8 Million in 2024, it is projected to reach US$ 825.2 Million by 2033.
As per our report Western Europe Pulmonary Pressure Monitors Market, the market size is valued at US$ 493.8 Million in 2024, projecting it to reach US$ 825.2 Million by 2033. This translates to a CAGR of approximately 5.9% during the forecast period.
The Western Europe Pulmonary Pressure Monitors Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Western Europe Pulmonary Pressure Monitors Market report:
The Western Europe Pulmonary Pressure Monitors Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Western Europe Pulmonary Pressure Monitors Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Western Europe Pulmonary Pressure Monitors Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)