Today, mAB treatment for people suffering from COVID-19 approved by the FDA intended for emergency use reveals that it proves successful in reducing the chances of severe diseases, hospitalization, and death by almost 70% by shortening hospital stays. Apart from that, coverage of mAB products for treating COVID-19 is another contributing factor for the growth of this market during the onset of the pandemic. Furthermore, the National Institute for Health and Care Excellence (NICE) updates managing COVID-19 guidelines with monoclonal antibody recommendations. For records, in October 2021, the new advice recommended offering a combination of "casirivimab" and "imdevimab" for treating COVID-19 patients aged 12 and above who are in hospital. Moreover, both the products are licensed for the prophylaxis and treatment of acute COVID-19 infection.
The European market is severely hit due to the exponential increase of COVID-19 cases in the region. Many nations are revealing more patients per day now than during the first wave earlier this year. Lockdowns are being reintroduced in UK, Spain, and Italy, and Ireland, as per the European Centre for Disease Prevention and Control (ECDC). Due to the second wave of COVID-19 crises, the government of many nations is heading towards mounting up testing capacity. Countries in the European regions were profoundly affected due to the COVID-19 pandemic. For instance, countries such as Italy, Spain, and France have recorded the most significant number of positive cases and have registered the maximum number of deaths. The rising rate of coronavirus results in increased stress on the region's healthcare system, raising the demand for diagnostic tests in its healthcare system, supporting the expansion of the sector in this region. Moreover, In December 2021, Pfizer and BioNTech announced that the companies will amend the clinical safety study while evaluating the safety, tolerability, and immunogenicity of the Pfizer-BioNTech COVID-19 vaccine in children 6 months to under 5 years of age.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the monoclonal antibodies market. The Europe monoclonal antibodies market is expected to grow at a good CAGR during the forecast period.
Europe Monoclonal Antibodies Market Segmentation
By Country
Companies Mentioned
| Report Attribute | Details |
|---|---|
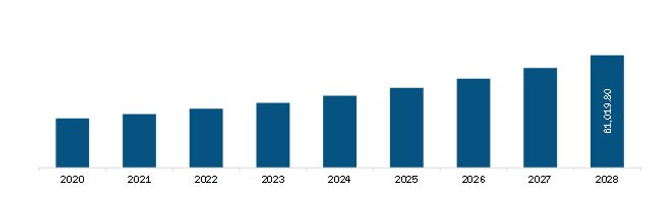
| Market size in 2021 | US$ 29,254.3 Million |
| Market Size by 2028 | US$ 61,019.8 Million |
| CAGR (2021 - 2028) | 11.1% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Source
|
|
Regions and Countries Covered
|
|
| Europe | UK, Germany, France, Russia, Italy, Rest of Europe |
| Market leaders and key company profiles |
|

The Europe Monoclonal Antibodies Market is valued at US$ 29,254.3 Million in 2021, it is projected to reach US$ 61,019.8 Million by 2028.
As per our report Europe Monoclonal Antibodies Market, the market size is valued at US$ 29,254.3 Million in 2021, projecting it to reach US$ 61,019.8 Million by 2028. This translates to a CAGR of approximately 11.1% during the forecast period.
The Europe Monoclonal Antibodies Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Monoclonal Antibodies Market report:
The Europe Monoclonal Antibodies Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Monoclonal Antibodies Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Monoclonal Antibodies Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)