The availability of technologically advanced products and treatment services allows surgeons, physicians, and other healthcare providers to offer superior services to their patients, which helps them maintain the optimum quality of life. The innovations are not limited to healthcare facilities, and they are extended to programs that are being designed for patients to get benefits out of treatment facilities. Heart diseases are complex, and the success of their intervention depends on innovations bringing treatment options that were deemed impossible earlier into reality. The heart disease treatment landscape—from structural inventions to surgical valve therapies—has experienced breakthrough innovations over the past few decades. Companies in the structural heart market offer a broad portfolio of solutions for different heart conditions, including vascular disease, irregular heartbeats, and heart valve diseases. Physicians can now manage arrhythmias by using remote monitoring devices to keep a tab on their patients, thereby treating an array of structural heart conditions.
The food and Drug Administration (FDA) have approved various therapies that are limited to the selected groups the patients. For instance, Premier Health, one of the providers of innovative structural heart services, offers alcohol septal ablation treatment for hypertrophic cardiomyopathy. The treatment mainly uses a catheter-based technology. The balloon valvuloplasty, uses a transcatheter approach for widening up stiff and narrow heart valves, atrial septal defect transcatheter repair which closes an opening between the heart’s left and right atrium without surgery and such more services are being provided by the service providers. CentraCare's Heart & Vascular Center used EchoPixel's True3D and HTG pre-planning software, which provides mixed reality capabilities that allow structural heart care teams to interact with a real-time holographic version of the patient's anatomy without physically touching it or exposing it to contrast agents and radiations.
The innovative technologies and techniques of structural heart programs are specifically designed to provide quality care for the best results for patients and provides less complications and shorter recovery times with long-lasting advantages. Such technologies are likely to attract more patients suffering from the structural heart diseases and eventually the benefits of the services are likely to enhance the growth of the market in the forecasted period.
The European structural heart market is segmented into Germany, the UK, France, Italy, Spain, and the Rest of Europe. The region holds a significant market share in the structural heart market. The European structural heart market is expected to witness significant growth during the forecast period due to factors such as significant growth in elderly population, rise in the incidence of CVDs, and launch of innovative products by market players in the region. Also, there has been an increase in the number of product approvals for treating heart disease in Europe. Moreover, market players have undertaken a number of initiatives to attract a larger customer base and sustain the competitive nature of the market. All these factors are fueling the market growth in the region.
Europe Structural Heart Market Segmentation
The Europe structural heart market is segmented int product, procedure, end user, and country.
Based on product, the Europe structural heart market is segmented into occluders, annuloplasty rings, heart valve balloons, and others. The occluders segment held the largest share of the Europe structural heart market in 2022.
Based on procedure, the Europe structural heart market is segmented into heart valve stenosis, heart valve regurgitation, and left atrial appendage closure. The heart valve stenosis segment held the largest share of the Europe structural heart market in 2022.
Based on end user, the Europe structural heart market is segmented into hospitals, ambulatory surgical centers, and cardiac centers. The hospitals segment held the largest share of the Europe structural heart market in 2022.
Based on country, the Europe structural heart market is segmented into Germany, the UK, France, Italy, Spain, and the Rest of Europe. Germany dominated the Europe structural heart market in 2022.
Abbott Laboratories; Medtronic Plc; Boston Scientific Corp; Artivion Inc; Edwards Lifesciences Corp; Lepu Medical Technology Beijing Co Ltd; Braile Biomedical Industry, Commerce and Representations; and Micro Interventional Devices Inc are some of the leading companies operating in the Europe structural heart market.

| Report Attribute | Details |
|---|---|
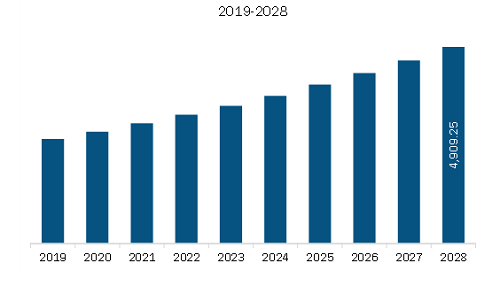
| Market size in 2022 | US$ 3,213.80 Million |
| Market Size by 2028 | US$ 4,909.25 Million |
| CAGR (2022 - 2028) | 7.3% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2028 |
| Segments Covered |
By Product
|
|
Regions and Countries Covered
|
|
| Europe | UK, Germany, France, Russia, Italy, Rest of Europe |
| Market leaders and key company profiles |
|

The Europe Structural Heart Market is valued at US$ 3,213.80 Million in 2022, it is projected to reach US$ 4,909.25 Million by 2028.
As per our report Europe Structural Heart Market, the market size is valued at US$ 3,213.80 Million in 2022, projecting it to reach US$ 4,909.25 Million by 2028. This translates to a CAGR of approximately 7.3% during the forecast period.
The Europe Structural Heart Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Structural Heart Market report:
The Europe Structural Heart Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Structural Heart Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Structural Heart Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)