Due to advancements in technologies related to feasibility and processing times, circulating tumor cell diagnostics and liquid biopsy have recently gathered considerable interest as a noninvasive alternative to tissue biopsy in cancer patients. The implementation of liquid biopsy procedures is supported by a greater understanding of improved minimally invasive cancer detection technologies. The market growth will be influenced by the rising demand for noninvasive tools, and laboratory acceptance of CTCs tests and liquid biopsy. A liquid biopsy allows doctors to obtain information about tumors from a simple blood sample. A blood test is painless, noninvasive, cost-effective, and less time-consuming. In addition, CTCs, cfDNAs, exosomes, and microvesicles can be detected in a blood sample, making blood-based liquid biopsies more popular.
The COVID-19 emergency has encouraged the general population to try out digital services and scale up their usage, particularly to avoid having to visit a health center in person. The unprecedented impact of COVID-19 has been far-reaching, but the effects on cancer patients make them one of the worst affected groups. COVID-19 has profoundly impacted the number of patients undergoing cancer screening, diagnosis, and treatment. The increased pressure due to the growing rate of COVID-19 patients' hospitalization led to the re-profiling of many hospitals and departments, including oncology clinics, for accommodating the increasingly large number of COVID-19 patients. According to one study, many diagnostic and treatment procedures, including 2.3 million cancer surgeries, were canceled or postponed worldwide. Thus, the pandemic hindered the circulating tumor cell (CTC) diagnostics market growth.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the circulating tumor cell (CTC) diagnostics market. The Europe circulating tumor cell (CTC) diagnostics market is expected to grow at a good CAGR during the forecast period.
Europe Circulating Tumor Cell (CTC) Diagnostics Market Segmentation
By Technology
| Report Attribute | Details |
|---|---|
| Market size in 2021 | US$ 508.0 Million |
| Market Size by 2028 | US$ 957.8 Million |
| CAGR (2021 - 2028) | 9.5% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Technology
|
|
Regions and Countries Covered
|
|
| Europe | UK, Germany, France, Russia, Italy, Rest of Europe |
| Market leaders and key company profiles |
|

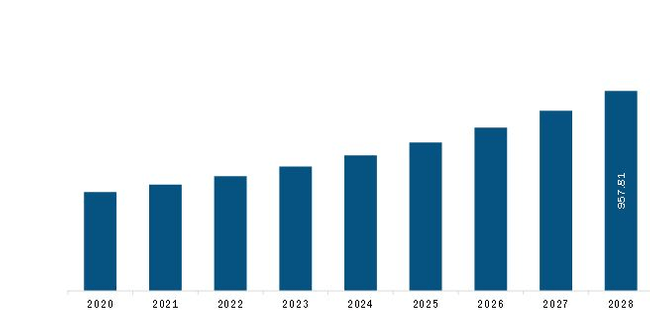
The Europe Circulating Tumor Cell (CTC) Diagnostics Market is valued at US$ 508.0 Million in 2021, it is projected to reach US$ 957.8 Million by 2028.
As per our report Europe Circulating Tumor Cell (CTC) Diagnostics Market, the market size is valued at US$ 508.0 Million in 2021, projecting it to reach US$ 957.8 Million by 2028. This translates to a CAGR of approximately 9.5% during the forecast period.
The Europe Circulating Tumor Cell (CTC) Diagnostics Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Circulating Tumor Cell (CTC) Diagnostics Market report:
The Europe Circulating Tumor Cell (CTC) Diagnostics Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Circulating Tumor Cell (CTC) Diagnostics Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Circulating Tumor Cell (CTC) Diagnostics Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)