Improving the availability of safe and adequate blood for transfusion procedures should be an integral part of every country’s national health care policy and infrastructure. The need for blood transfusion may arise anytime in urban and rural areas. In the past, the unavailability of blood has led to deaths for many patients suffering from ill-health. An adequate and reliable supply of safe blood can be assured by having stable facilities for regular, voluntary, and unpaid blood donors. They are also the safest group of donors as the prevalence of blood-borne infections is lowest among these donors. The most common surgeries that require blood transfusion include cardiovascular procedures, C-section procedures, and injuries. Moreover, due to natural calamities and widespread causalities, the demand for blood transfusion is increasing. A surge in the incidence of blood disorders such as anemia, erythrocytosis, leukocytosis, leukopenia, and thrombocytosis is also boosting the blood filter market. As per WHO, most of the children with thalassemia are born in low-income countries, where access to blood transfusion is either difficult or is available to a small percentage of the population. Blood filters used in transfusion procedures are woven mesh made from polyester or nylon; these are screen-type filters placed between the product bag and the patient. These filters are designed to protect the patients from potentially harmful macro-aggregates such as platelets, leucocytes, and fibrin proteins—and non-blood component particulate matter. In the majority of developed countries, the blood transfusion filters are provided by governments or community-based programs. Thus, the availability of suitable facilities for blood donation and voluntary donor drives the number of blood transfusion procedures, which boosts the Europe blood filler market growth.
European countries were hit due to the exponential increase in COVID-19 cases. According to the European Center for Disease Prevention and Control (ECDC), social restrictions were reintroduced in the UK, Spain, Italy, and Ireland after the beginning of the second wave of pandemic in these countries. Spain, Italy, Germany, France, and the UK are among the worst-hit European countries. The blood filter market stakeholders were losing their businesses due to the temporary shutdown of industries in the region. In 2019, the European Commission's (EC) assessment of the EU Blood Directive concluded that current regulations are insufficient to ensure an adequate and sustainable blood supply. According to the European Blood Alliance and the ECDC, European national and regional blood services reported an average decrease of 9% (median, range 1–27%) in blood and blood components collected in March and April 2020 compared to the corresponding period in 2019. As a result, there was an overall decrease of 12% in blood components distributed to hospitals (range 1–18%). At the beginning of the pandemic, several centers in Italy, Spain, and Greece reported a 32%, 11–49%, and 25–60% decrease in. In the UK, the RBC units received by blood collection centers dropped by ~30% in March 2020, which has leveled off at ~10% by May 2020. The demand for transfusions was further decreased due to delays in cancer treatments, and reduced road accidents and subsequent trauma cases. In 2019, the Seraph 100 Microbind Affinity Blood Filter from ExThera Medical Corporation was approved for its sale in Europe. The product was approved by the FDA for emergency use in COVID-19 patients admitted to the intensive care unit with confirmed or imminent respiratory failure. The blood is cleaned with dialysis by pumping it through the filter. The Seraph is indicated for reducing pathogens in bloodstream, such as SARS-CoV-2, MRSA, and other drug-resistant bacteria.
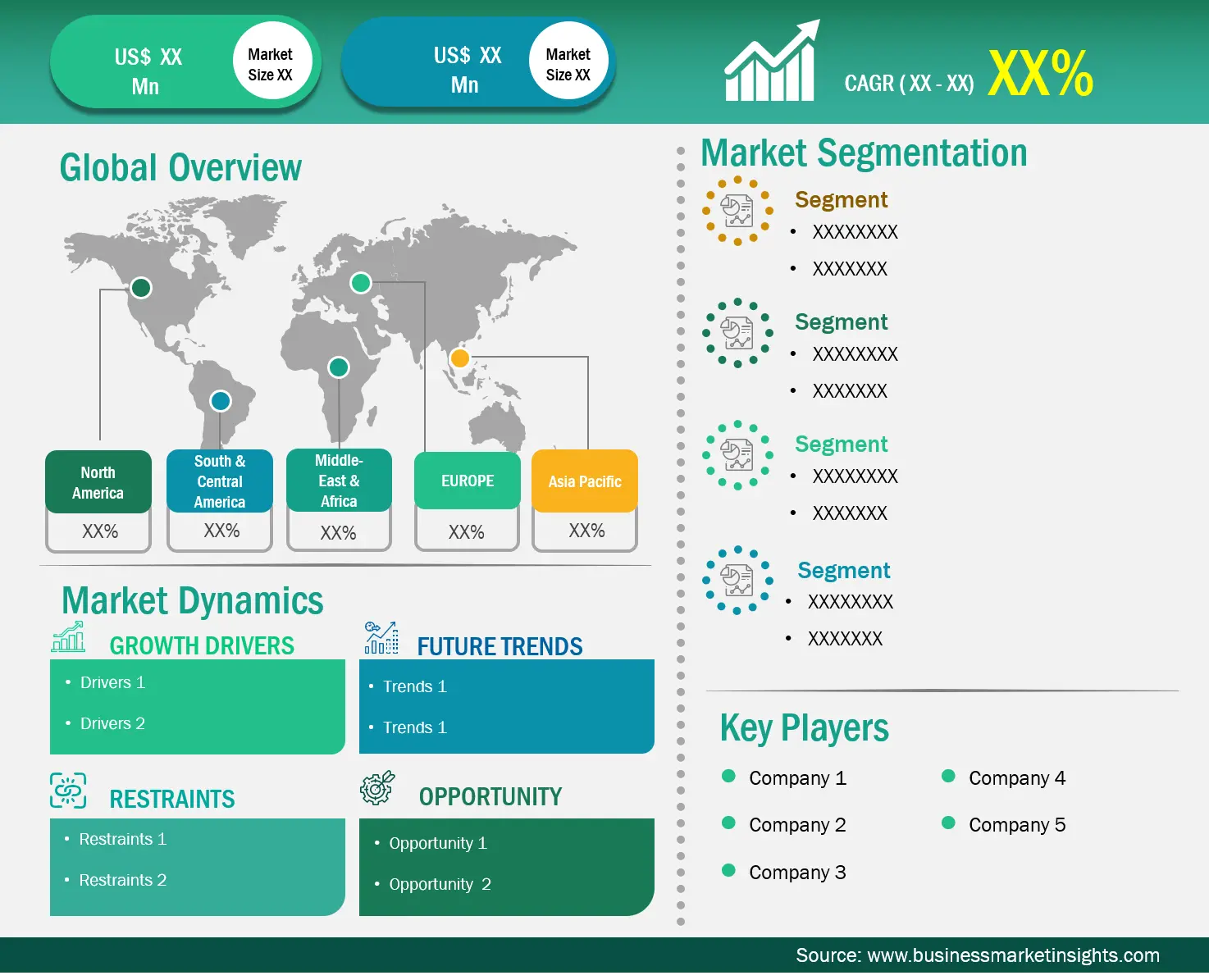
| Report Attribute | Details |
|---|---|
| Market size in 2021 | US$ 1,12,166.63 thousand |
| Market Size by 2028 | US$ 1,45,156.91 thousand |
| CAGR (2021 - 2028) | 3.8% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Product
|
|
Regions and Countries Covered
|
|
| Europe | UK, Germany, France, Russia, Italy, Rest of Europe |
| Market leaders and key company profiles |
|

The Europe Blood Filter Market is valued at US$ 1,12,166.63 thousand in 2021, it is projected to reach US$ 1,45,156.91 thousand by 2028.
As per our report Europe Blood Filter Market, the market size is valued at US$ 1,12,166.63 thousand in 2021, projecting it to reach US$ 1,45,156.91 thousand by 2028. This translates to a CAGR of approximately 3.8% during the forecast period.
The Europe Blood Filter Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Blood Filter Market report:
The Europe Blood Filter Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Blood Filter Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Blood Filter Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)