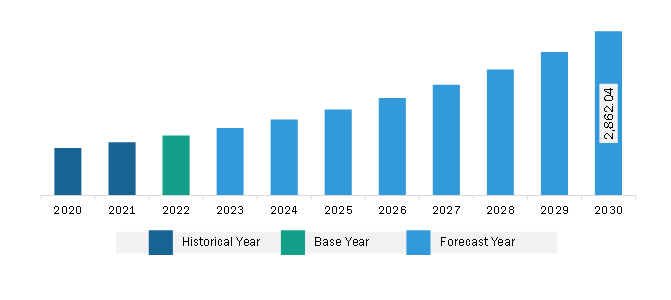
The North America vaccine adjuvants market was valued at US$ 1,044.84 million in 2022 and is expected to reach US$ 2,862.04 million by 2030; it is estimated to register a CAGR of 13.4% from 2022 to 2030.
According to the World Bank 2023 report, bacterial, viral, parasite, or fungal diseases can be contagious, spreading directly from person to person or animal to person. As per the National Institute of Health (NIH) report, the H1N1 influenza pandemic resulted in losses of ~US$ 2.8 billion to the Mexican tourism industry, as the footfall of tourists decreased by nearly 1 million over a 5-month window of the contagious spread of H1N1 virus. The Mexican state of Texcoco reported an outbreak of low pathogenicity avian influenza (LPAI) A(H5N2) in March 2024, and Temascalapa municipality in the same state reported another outbreak in April. Additionally, in the state of Michoacán, a private poultry farm detected a high-pathogenicity avian influenza A(H5N2) outbreak that was discovered in March 2024. In 2022, it was reported that both LPAI and HPAI A(H5) subtypes have been found in bird species in Mexico, both recently and in the past. Although they typically infect animals, animal influenza viruses can also infect people. Human infections have mostly been contracted via close contact with animals that are infected or contaminated environments. Influenza A viruses can be categorized as avian influenza, swine influenza, or other animal influenza viruses based on their original host. Therefore, the emergence of various infectious diseases with the associated cost burden accelerates the manufacturers' demand for vaccines and vaccine adjuvants.
COVAXIN, a vaccine developed by Bharat Biotech to fight against the COVID-19 pandemic, uses Alhydroxiquim-II as an adjuvant. The biotech company ViroVax LLC discovered and tested this adjuvant in the laboratory. The pharmacological action of Alhydroxiquim-II is that it travels through lymph nodes and activates TLR7 and TLR8 cellular receptors, which play a vital role in the immune response against viruses. Alhydroxiquim-II is the first adjuvant responsible for activating TLR7 and TLR8, thus stimulating people’s immune responses. Therefore, the rising number of infectious disease outbreaks and pandemics, accounting for high healthcare costs, propels the demand for vaccine adjuvants.
The North America vaccine adjuvants market has been segmented into the US, Canada, and Mexico. Fast product approval processes benefit the vaccine adjuvants market in the US. In May 2023, GlaxoSmithKline (GSK) won US Food and Drug Administration (FDA) approval for its Arexvy (respiratory syncytial virus vaccine, adjuvanted) vaccine intended for the prevention of lower respiratory tract disease (LRTD) caused by respiratory syncytial virus (RSV). Thus, Arexvy became the world's first FDA-approved RSV vaccine for older adults. In October 2022, Novavax, Inc. received an Emergency Use Authorization (EUA) from the FDA for its COVID-19 adjuvanted vaccine, indicated as a booster for adult patients.
 North America Vaccine Adjuvants Market Segmentation
North America Vaccine Adjuvants Market Segmentation
The North America vaccine adjuvants market is categorized into adjuvant class, type, and country.
Based on adjuvant class, the North America vaccine adjuvants market is segmented into mineral salt adjuvant, emulsion adjuvant, liposome adjuvant, and others. The mineral salt adjuvant segment held the largest share of the North America vaccine adjuvants market share in 2022.
By type, the North America vaccine adjuvants market is bifurcated into human vaccine adjuvant and veterinary vaccine adjuvant. The human vaccine adjuvant segment held a larger share of North America vaccine adjuvants market in 2022.
By country, the North America vaccine adjuvants market is segmented into the US, Canada, and Mexico. The US dominated the North America vaccine adjuvants market share in 2022.
Croda International Plc; CSL Ltd; Dynavax Technologies Corp; GSK Plc; Hawaii Biotech Inc; InvivoGen SAS; Novavax Inc; Phibro Animal Health Corp; Seppic SA; and SPI Pharma Inc are some of the leading companies operating in the North America vaccine adjuvants market.

| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 1,044.84 Million |
| Market Size by 2030 | US$ 2,862.04 Million |
| CAGR (2022 - 2030) | 13.4% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2030 |
| Segments Covered |
By Adjuvant Class
|
|
Regions and Countries Covered
|
|
| North America | US, Canada, Mexico |
| Market leaders and key company profiles |
|
The North America Vaccine Adjuvants Market is valued at US$ 1,044.84 Million in 2022, it is projected to reach US$ 2,862.04 Million by 2030.
As per our report North America Vaccine Adjuvants Market, the market size is valued at US$ 1,044.84 Million in 2022, projecting it to reach US$ 2,862.04 Million by 2030. This translates to a CAGR of approximately 13.4% during the forecast period.
The North America Vaccine Adjuvants Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Vaccine Adjuvants Market report:
The North America Vaccine Adjuvants Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Vaccine Adjuvants Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Vaccine Adjuvants Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)