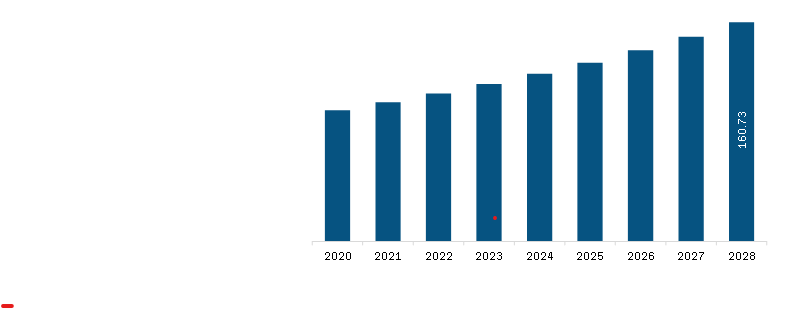
The North America pulmonary function testing systems market was valued at US$ 108.42 million in 2022 and is expected to reach US$ 160.73 million by 2028; it is estimated to register a CAGR of 6.8% from 2022 to 2028.
Rising Incidence of Lung-Related Disorders Fuels North America Pulmonary Function Testing Systems Market
The PFT systems provide accurate results, particularly lung volume, capacity, rates of flow, and gas exchange. PFT systems utilization assists in assessing treatment for asthma, emphysema, and other chronic lung problems. Through the assessment of lung volumes, capacities, rates of flow, and gas exchange, PFTs can provide information that can be evaluated by physicians to help diagnose certain lung disorders effectively and decide the right treatment for patients. Further, PFTs are noninvasive procedures and are usually recommended by physicians for patients with no hospital stay. Spirometry is a type of commercial pulmonary function testing system. Spirometry testing is useful for determining if the patient is suffering from lung disease, type of lung disease, progression of the lung disease, and impairment of lung disease. Spirometry measures airflow and can evaluate a broad range of lung diseases among patients. Thus, the rising incidence of lung-related disorders is fueling the use of pulmonary function testing systems in North America.
North America Pulmonary Function Testing Systems Market Overview
The North America pulmonary function testing systems market is segmented into the US, Canada, and Mexico. The US held the largest share of the North America pulmonary function testing systems market in 2022. The North America pulmonary function testing systems market growth is attributed to the high implementation of portable systems and a rise in the usage of respiratory devices among the physician groups (clinics) in the region.
North America Pulmonary Function Testing Systems Market Revenue and Forecast to 2028 (US$ Million)
North America Pulmonary Function Testing Systems Market Segmentation
The North America pulmonary function testing systems market is segmented on the basis of type, test type, component, application, end user, and country.
Based on type, the North America pulmonary function testing systems market is bifurcated into cart-based PFT systems and stationery PFT systems. The cart-based PFT systems segment held a larger share of the North America pulmonary function testing systems market in 2022.
Based on test type, the North America pulmonary function testing systems market is segmented into spirometry, lung volume, gas exchange testing, maximum voluntary ventilation, oxygen titration test, and others. The spirometry segment held the largest share of the North America pulmonary function testing systems market in 2022.
Based on component, the North America pulmonary function testing systems market is segmented into hardware, software, and services. The hardware segment held the largest share of the North America pulmonary function testing systems market in 2022.
Based on application, the North America pulmonary function testing systems market is segmented into chronic obstructive pulmonary disorder, asthma, chronic shortness of breath, restrictive lung disease, and others. The chronic obstructive pulmonary disorder segment held the largest share of the North America pulmonary function testing systems market in 2022.
Based on end-user, the North America pulmonary function testing systems market is segmented into hospitals, specialty clinics, and diagnostic laboratories. The hospitals segment held the largest share of the North America pulmonary function testing systems market in 2022.
Based on country, the North America pulmonary function testing systems market is segmented into the US, Canada, and Mexico. The US dominated the share of the North America pulmonary function testing systems market in 2022.
COSMED Srl, KoKo PFT Ltd, MGC Diagnostics Corp, Morgan Scientific Inc, ndd Medical Technologies Inc, PulmOne Advanced Medical Devices Ltd, Schiller AG, and Vyaire Medical Inc are the leading companies operating in the North America pulmonary function testing systems market.

| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 108.42 Million |
| Market Size by 2028 | US$ 160.73 Million |
| CAGR (2022 - 2028) | 6.8% |
| Historical Data | 2020-2021 |
| Forecast period | 2023-2028 |
| Segments Covered |
By Type
|
|
Regions and Countries Covered
|
|
| North America | US, Canada, Mexico |
| Market leaders and key company profiles |
|

The North America Pulmonary Function Testing Systems Market is valued at US$ 108.42 Million in 2022, it is projected to reach US$ 160.73 Million by 2028.
As per our report North America Pulmonary Function Testing Systems Market, the market size is valued at US$ 108.42 Million in 2022, projecting it to reach US$ 160.73 Million by 2028. This translates to a CAGR of approximately 6.8% during the forecast period.
The North America Pulmonary Function Testing Systems Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the North America Pulmonary Function Testing Systems Market report:
The North America Pulmonary Function Testing Systems Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The North America Pulmonary Function Testing Systems Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the North America Pulmonary Function Testing Systems Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)