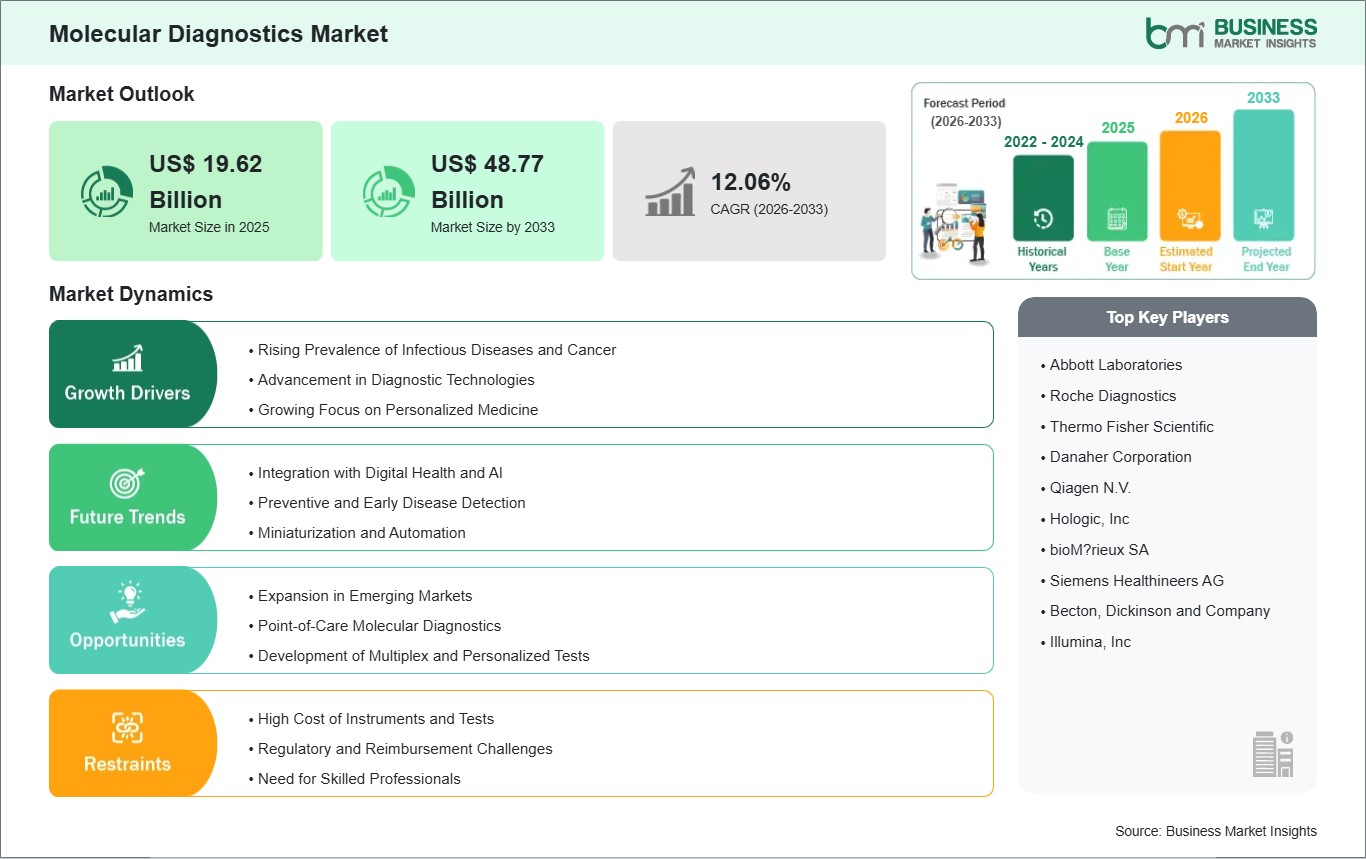
The Molecular Diagnostics Market size is expected to reach US$ 48.77 Billion by 2033 from US$ 19.62 Billion in 2025. The market is estimated to record a CAGR of 12.06% from 2026 to 2033.
The global molecular diagnostics market experiences a major shift as healthcare systems across the globe put more focus on early and accurate disease detection in order to improve patient outcomes and cope with the increasing disease burdens. The market is flooded with demands for rapid and accurate diagnostic solutions for infectious diseases, oncology, genetic disorders, and chronic conditions, which is driven by clinical necessity and the growth of personalized medicine. The mentioned technologies such as PCR, next, generation sequencing (NGS), and isothermal amplification are at the center of this change as they allow very sensitive and specific detection of pathogens and genetic markers.
The rising occurrence of infectious diseases such as tuberculosis, HIV, and newly emerging pathogens, in combination with the focus on early detection of cancer, is the main reason for the strong uptake of molecular testing platforms. On top of that, regulatory initiatives that support the market, growing healthcare infrastructure in developing countries, and increasing investment in advanced diagnostic research are some of the factors that contribute to the market growth. In terms of regions, North America is still holding a leading position due to strong healthcare infrastructure and the widespread adoption of advanced diagnostics while the Asia Pacific region becomes the fastest, growing one because of increasing healthcare expenditure and growing biotech sectors. Two of the most talked, about points in the future of molecular diagnostics are point, of, care testing and digital health initiatives that will improve accessibility and clinical utility.
Key segments that contributed to the derivation of the Molecular Diagnostics market analysis are component, technology, application, and end user.
Due to an increase in the demand for accurate and timely detection of disease, there is a rapid evolution in how healthcare services are provided globally. Molecular diagnostics allow for the accurate identification of specific pathogens and pathological changes (e.g., genetic changes) which aid in diagnosing a specific disease or abnormality early in the disease course. Early diagnosis with molecular techniques has proven critical to improving patient outcomes particularly within the fields of oncology, infectious diseases, inherited genetic disorders and is essential for developing successful treatment protocols for patients diagnosed with these conditions. The rapid increase in chronic and infectious diseases globally, including cancer, tuberculosis, HIV, and emerging virus outbreaks, coupled with the shift toward personalized medicine and the use of targeted therapies, are creating an urgent need for reliable molecular testing platforms. Hospitals, clinics and diagnostic laboratories are developing and implementing innovative techniques such as polymerase chain reactions, next-generation sequencing, and isothermal amplification to address the need for reliable molecular testing solutions to support personalized medicine and targeted therapies. There is an increasing emphasis on preventive care and early intervention from both healthcare providers and policymakers and, as a result, molecular diagnostics represent a critical component for improving the health of a population, decreasing future healthcare costs, and improving decision-making in clinical practice.
The growth in use of point-of-care (POC) diagnostic solutions is fueling the growth of the Molecular Diagnostics Market. The introduction of technology-driven innovations which are smaller, faster, and better to use have led to the creation of POC molecular testing systems that are deployable outside traditional laboratory environments (e.g. clinics, rural clinics, and even in low-resource locations). Timely diagnosis of disease provides an essential tool to clinicians regarding decision-making for immediate clinical intervention regarding infectious disease outbreaks and monitoring patients for cancer therapy. While many of the world’s emerging economies (e.g. Asia-Pacific, Latin America, and Africa) are benefiting from increased healthcare spending, developed medical infrastructure, and increased familiarity with molecular testing, these regions are ripe for increased growth in the molecular diagnostics market. The increased incidence of infectious diseases, as well as ongoing government support in these regions for enhancing diagnostic capabilities, are aiding in-creased adoption of molecular testing in these areas. Furthermore, there is an increasing interest and demand for lower cost and higher accuracy POC molecular tests that can expand into untapped markets due to the availability of new technologies. Establishing business relationships with local healthcare providers, investing in mobile/portable diagnostic systems, and developing culturally and regionally specific testing procedures and products are additional areas for growth and potential market entry. Leveraging trends in the above categories will allow companies to broaden their brand awareness, increase access to healthcare services, and support long-term growth within the Market.
By component, the molecular diagnostics market is segmented into instruments, reagents and kits. The reagents and kits segment dominated the market in 2024. The demand for reagents and kits is largely a consequence of the increasing number of diagnostic tests and the growing adoption of advanced molecular techniques. As healthcare providers keep on expanding testing for infectious diseases, oncology, and genetic disorders, consumables are perpetually required, thus this segment becomes the main market driver.
By technology, the market is segmented into polymerase chain reaction, next-generation sequencing (NGS) / DNA sequencing, in situ hybridization, DNA microarrays, others. The polymerase chain reaction segment held the largest share of the market in 2024. Rapid, accurate, and reliable diagnostic results are the main factors behind PCR adoption. In addition, its versatility across different applications, low price, and established clinical use make it the most suitable test for both routine and specialized testing.
By application, the market is segmented into infectious diseases, oncology, genetic disorders, companion diagnostics, others. The infectious diseases segment held the largest share of the market in 2024. The prevalence of infectious diseases globally and the need for early detection as well as accurate identification of pathogens are the major factors leading to the growth of this segment.
By end user, the market is segmented hospitals and clinics, diagnostic laboratories, academic and research institutes, others. The hospitals and clinics segment held the largest share of the market in 2024. the rise in patient numbers, the need for fast diagnosis, and the growth of hospitals in both developed and emerging markets are the major factors that lead to the increase of this segment.
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 19.62 Billion |
| Market Size by 2033 | US$ 48.77 Billion |
| Global CAGR (2026 - 2033) | 12.06% |
| Historical Data | 2022-2024 |
| Forecast period | 2026-2033 |
| Segments Covered | By Component
|
|
Regions and Countries Covered
| |
| North America | US, Canada, Mexico |
| Europe | Belgium, Austria, Finland, Denmark, Greece, Poland, Romania, Russia, Ukraine, Czech Republic, Slovakia, Bulgaria, Italy, Luxembourg, Germany, Switzerland, France, Netherlands, Norway, Portugal, Spain, Sweden, United Kingdom |
| Asia-Pacific | Australia, China, India, Japan, South Korea, Indonesia, Malaysia, Philippines, Singapore, Thailand, Vietnam, Bangladesh, New Zealand, Taiwan |
| South and Central America | Brazil, Argentina, Peru, Chile, Colombia |
| Middle East and Africa | Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates, Turkiye, South Africa, Egypt, Algeria, Nigeria |
| Market leaders and key company profiles |
|
The "Molecular Diagnostics Market Size and Forecast (2022–2033)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the Molecular Diagnostics market report is divided into five regions: North America, Asia Pacific, Europe, Middle East and Africa, and South and Central America. The Molecular Diagnostics market in Asia Pacific is expected to grow significantly during the forecast period.
The Asia-Pacific Molecular Diagnostics market is segmented into China, Japan, South Korea, India, Australia, New Zealand, Indonesia, Malaysia, the Philippines, Singapore, Thailand, Vietnam, Taiwan, Bangladesh, and the Rest of Asia. Increasing healthcare expenses, expanded infrastructure, and greater knowledge of advanced diagnostic technology has created a growing Asia Pacific market for molecular diagnostics. Countries such as China, India, Japan, South Korea, and Australia have experienced substantial growth in their utilization of molecular diagnostics for infectious disease testing as well as oncology, genetic disorders and personalized medicine. Infectious disease continues to pose large public health threats, with high incidence rates of tuberculosis, hepatitis, and HIV. The growing incidence of cancer and other genetic diseases is creating an urgent need for timely and accurate diagnosis, resulting in continued growth within this sector. Government initiatives to improve healthcare delivery, enhance the ability of health services to monitor disease, and create early detection programs have all contributed to increased utilization of molecular diagnostics. Many private diagnostic laboratories, hospitals and medical centres now have access to state-of-the-art molecular testing platforms and are expanding the availability of these tests to patients, particularly in urban and semi-urban environments. Point-of-care rapid molecular diagnostic devices are gaining additional momentum and increasing accessibility to diagnostic testing for patients living in remote and resource constrained settings. New digital health tools, including telemedicine services and electronic health records, are improving efficiency of testing and overall patient management. While the Asia Pacific region is also facing some challenges related to regulatory variability and the sensitivity of patients to costs; there are many opportunities for continued growth because of the large patient population in the Asia Pacific region with rising income levels and greater acceptance of innovative health solutions.

The Molecular Diagnostics market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the molecular diagnostics market are:
The Molecular Diagnostics Market is valued at US$ 19.62 Billion in 2025, it is projected to reach US$ 48.77 Billion by 2033.
As per our report Molecular Diagnostics Market, the market size is valued at US$ 19.62 Billion in 2025, projecting it to reach US$ 48.77 Billion by 2033. This translates to a CAGR of approximately 12.06% during the forecast period.
The Molecular Diagnostics Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Molecular Diagnostics Market report:
The Molecular Diagnostics Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Molecular Diagnostics Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Molecular Diagnostics Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)