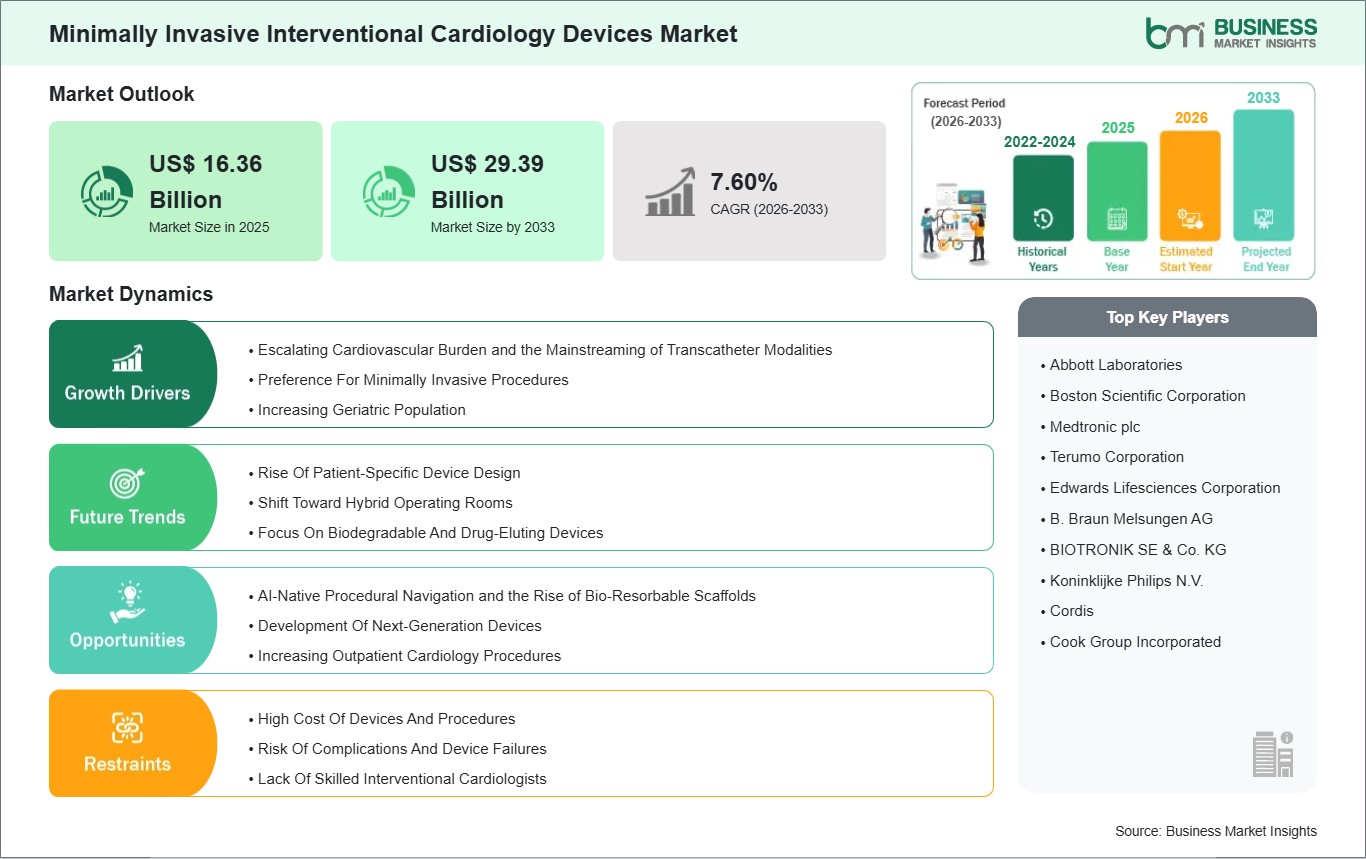
The Minimally Invasive Interventional Cardiology Devices Market size is expected to reach US$ 29.39 Billion by 2033 from US$ 16.36 Billion in 2025. The market is estimated to record a CAGR of 7.60% from 2026 to 2033.
Minimally invasive interventional cardiology devices refer to a specialized suite of medical instruments used to diagnose and treat structural and vascular heart conditions via small incisions rather than traditional open-heart surgery. Market expansion is fundamentally driven by the rising global prevalence of cardiovascular diseases (CVDs), such as coronary artery disease and heart failure, compounded by a global aging population susceptible to degenerative valve conditions. Furthermore, the shift toward outpatient cardiac care and the integration of robotic-assisted systems are enhancing procedural precision while reducing hospital stays. Government initiatives promoting early diagnosis and the expansion of specialized heart centers in emerging economies continue to bolster the steady adoption of these technologies.
However, several factors can restrain market growth. The high capital expenditure required for advanced imaging suites and robotic platforms can limit accessibility in resource-constrained regions. Stringent regulatory pathways and the need for extensive clinical evidence for high-risk Class III devices often prolong commercialization timelines. Additionally, the industry faces constraints due to pricing pressures and centralized procurement programs, particularly in markets such as China and parts of Europe, which can impact the profit margins of premium device manufacturers.
Despite these hurdles, the market holds significant opportunities in the development of bioresorbable vascular scaffolds and AI-enhanced intravascular imaging, which provide superior long-term safety profiles. The rise of transcatheter mitral and tricuspid therapies (TMTT) and the expansion of tele-interventional support, allowing experts to guide procedures remotely are expected to support long-term development within the sector.
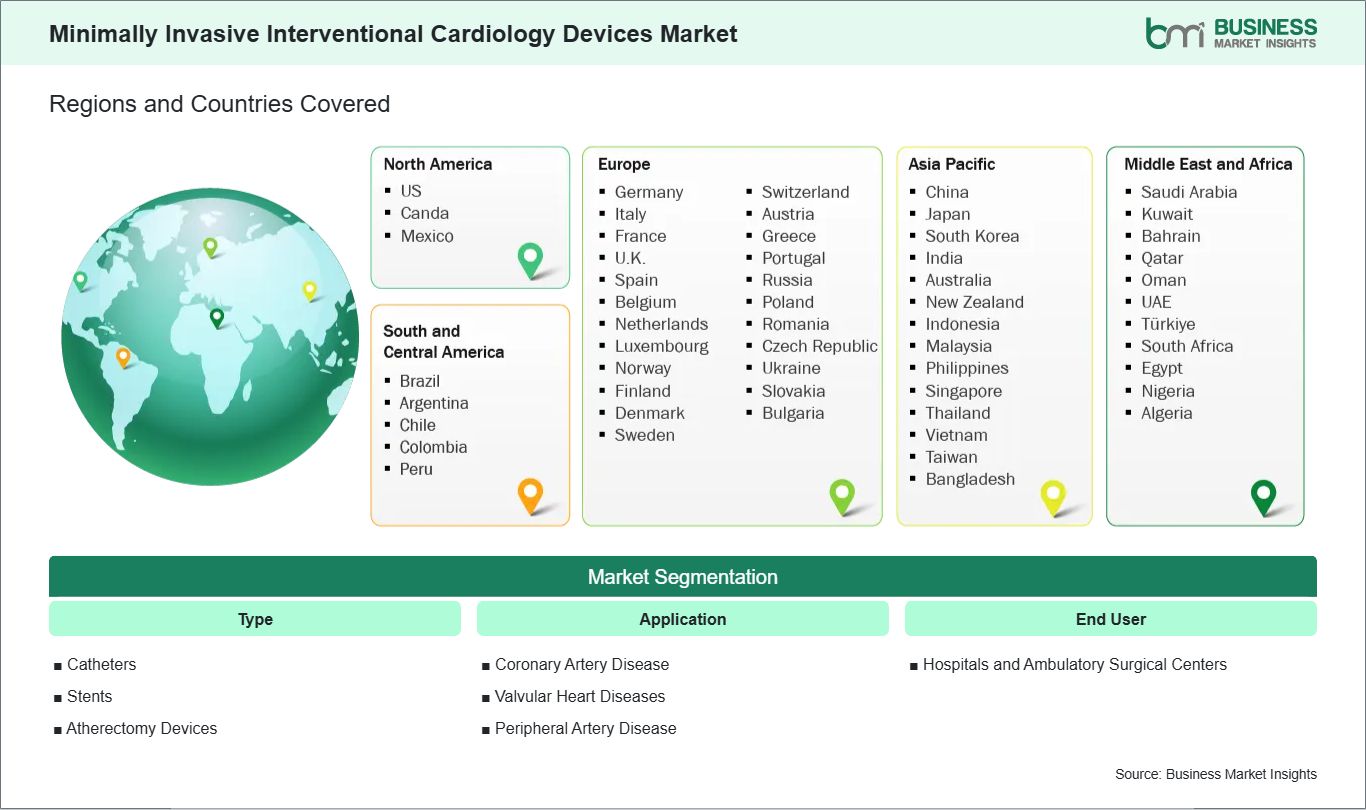
Key segments that contributed to the derivation of the Minimally Invasive Interventional Cardiology Devices market analysis are type, application, and end user.
The primary driver for the Minimally Invasive Interventional Cardiology Devices Market is the systemic global rise in chronic metabolic conditions and the aging of the global population. The Increasing Prevalence of Coronary Artery Disease and Aortic Stenosis acts as a foundational catalyst, as sedentary lifestyles and rising obesity rates necessitate a higher volume of percutaneous coronary interventions (PCI). This momentum is further propelled by the Strategic Shift Toward Transcatheter Aortic Valve Replacement (TAVR) in Low-Risk Patients; as clinical evidence continues to demonstrate non-inferiority to surgical replacement. Minimally invasive valve therapy is becoming the primary standard of care for a broader patient demographic. In the technological sphere, the Rapid Adoption of Intravascular Lithotripsy (IVL) and High-Resolution Imaging serves as a critical driver, enabling clinicians to treat heavily calcified lesions that were previously considered inaccessible via catheter-based methods. Furthermore, Favorable Reimbursement Frameworks for Ambulatory Surgical Centers (ASCs) are decentralizing procedural access, allowing for high-efficiency, cost-effective interventional care outside of traditional hospital settings. Together, these factors, disease incidence, the TAVR revolution, and procedural decentralization, ensure a robust and essential growth path for the global Minimally Invasive Interventional Cardiology Devices Market.
A significant high-value opportunity lies in the convergence of Interventional Hardware with Artificial Intelligence (AI) and Digital Twin Modeling. Next-generation platforms are being developed to utilize AI for real-time angiography analysis and automated plaque characterization, which significantly enhances the accuracy of stent sizing and reduces the likelihood of geographical miss. There is also a major growth frontier in the development of Advanced Bio-Resorbable Vascular Scaffolds (BVS); as manufacturers refine polymer science to ensure predictable resorption rates, these devices offer a disappearing solution that leaves the native vessel intact, potentially revolutionizing long-term outcomes in younger patients. Furthermore, the expansion of Pulsed Field Ablation (PFA) and Robotic-Assisted PCI presents a lucrative opportunity, offering sub-second energy delivery and sub-millimeter precision that minimizes physician radiation exposure and improves procedural repeatability. Beyond coronary care, the rise of Transcatheter Mitral and Tricuspid Therapies (TMVR/TTVR) offers a unique frontier, targeting a massive, underserved population with structural heart defects. Manufacturers who focus on Ultra-Low-Profile Delivery Systems and those pioneering Sustainable, Non-Plastic Coating Technologies are positioned to lead the global Minimally Invasive Interventional Cardiology Devices Market.
Based on the type, the Stents subsegment holds the primary market presence, acting as the ubiquitous standard for maintaining vessel patency following angioplasty. These devices are indispensable for the market, maintaining a significant position due to the high volume of percutaneous coronary interventions performed globally. While Catheters and Atherectomy Devices provide critical support, with catheters serving as the essential delivery and diagnostic vehicle, they hold secondary positions in terms of total system utilization. A notable trend is the surge in the Drug-Eluting Stents (DES) and Intravascular Lithotripsy (IVL) Catheters subsegments, which are registering the highest pace of adoption. These technologies are becoming essential for Complex Plaque Management, where the move toward advanced drug formulations and sonic pressure waves allows for the effective treatment of resistant blockages with significantly lower rates of restenosis.
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 16.36 Billion |
| Market Size by 2033 | US$ 29.39 Billion |
| Global CAGR (2026 - 2033) | 7.60% |
| Historical Data | 2022-2024 |
| Forecast period | 2026-2033 |
| Segments Covered | By Type
|
|
Regions and Countries Covered
| |
| North America | US, Canada, Mexico |
| Europe | Belgium, Austria, Finland, Denmark, Greece, Poland, Romania, Russia, Ukraine, Czech Republic, Slovakia, Bulgaria, Italy, Luxembourg, Germany, Switzerland, France, Netherlands, Norway, Portugal, Spain, Sweden, United Kingdom |
| Asia-Pacific | Australia, China, India, Japan, South Korea, Indonesia, Malaysia, Philippines, Singapore, Thailand, Vietnam, Bangladesh, New Zealand, Taiwan |
| South and Central America | Brazil, Argentina, Peru, Chile, Colombia |
| Middle East and Africa | Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates, Turkiye, South Africa, Egypt, Algeria, Nigeria |
| Market leaders and key company profiles |
|
The "Minimally Invasive Interventional Cardiology Devices Market Size and Forecast (2022–2033)" report provides a detailed analysis of the market covering below areas:
The geographical scope of the Minimally Invasive Interventional Cardiology Devices market report is divided into five regions: North America, Asia Pacific, Europe, Middle East & Africa, and South & Central America.
North America maintains the primary market position, supported by an advanced clinical infrastructure, high healthcare expenditure, and the rapid adoption of breakthrough technologies such as artificial intelligence-integrated intravascular imaging and robotic-assisted surgical platforms. Europe represents a mature and highly regulated market where growth is sustained by a robust institutional focus on reducing hospital stay durations and an increasing clinical shift toward transcatheter aortic valve replacement (TAVR) as a standard of care. Asia Pacific is identified as the most rapidly advancing region, propelled by accelerating healthcare modernization, significant investments in specialized cardiac catheterization laboratories, and an expanding middle-class demographic with increasing access to sophisticated cardiovascular interventions. Market progression is further supported by a transition toward bio-resorbable scaffolds and ultra-thin-strut drug-eluting stents, which facilitate physiological vessel healing and minimize long-term restenosis. The increasing adoption of intravascular lithotripsy (IVL) systems, alongside the strategic expansion of specialized ambulatory surgical centers (ASCs), solidifies Asia Pacific as a vital region for the evolution and future scaling of the Minimally Invasive Interventional Cardiology Devices Market.
The Minimally Invasive Interventional Cardiology Devices market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the key developments in the Minimally Invasive Interventional Cardiology Devices market are:
The Minimally Invasive Interventional Cardiology Devices Market is valued at US$ 16.36 Billion in 2025, it is projected to reach US$ 29.39 Billion by 2033.
As per our report Minimally Invasive Interventional Cardiology Devices Market, the market size is valued at US$ 16.36 Billion in 2025, projecting it to reach US$ 29.39 Billion by 2033. This translates to a CAGR of approximately 7.60% during the forecast period.
The Minimally Invasive Interventional Cardiology Devices Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Minimally Invasive Interventional Cardiology Devices Market report:
The Minimally Invasive Interventional Cardiology Devices Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Minimally Invasive Interventional Cardiology Devices Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Minimally Invasive Interventional Cardiology Devices Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)