The market's growth is attributed to factors such as the rising prevalence of bacterial infections and increasing private and public partnerships of leading pharmaceutical players. However, the tedious and expensive process of developing antibiotics is expected to restrain the market's growth during the forecast period.
Antimicrobial resistance (AMR) has become a significant threat worldwide. Due to environmental factors such as exposure to antibiotics and spontaneous mutations, bacteria are becoming highly resistant to many antibiotics. Therefore, various strategies are designed to measure and control the spread of AMR. Several governments and healthcare authorities implement constructive plans to combat such critical situations. In 2017, the government of Germany announced the launch of the Global R&D Collaboration Hub to oppose AMR to focus on bridging gaps in the new developments. In January 2019, the UK government launched its strategy to deal with AMR. The UK's government has a 5-year national action plan to deal with antimicrobial resistance. Also, it has the vision to reduce AMR by 2040. The plan was developed across the governments, including Scotland, Wales, and Northern Ireland, and their agencies and administrations with the support of a range of stakeholders.
In April 2020, Venatorx Pharmaceuticals and the Global Antibiotic Research and Development Partnership (GARDP) announced a collaboration to accelerate the development of, and access to, cefepime-taniborbactam. It is a broad-spectrum beta-lactamase inhibitor that restores the activities of cefepime against carbapenem-resistant Enterobacterales (CRE) and carbapenem-resistant Pseudomonas aeruginosa (CRPA). However, it is an investigational combination of the fourth-generation antibiotic cefepime with taniborbactam. Under the collaboration, GARDP and Venatorx look forward to developing cefepime-taniborbactam. The combination is under a phase 3 cUTI trial. Additionally, clinical trials in adults with multidrug-resistant infections and clinical development activities and trials to enable cefepime-taniborbactam for children, including newborns with severe bacterial infections, are being conducted. Such activities by public and private partnerships are projected to offer lucrative opportunities for the growth of the carbapenem-based antibiotics market during the forecast period.
| Report Attribute | Details |
|---|---|
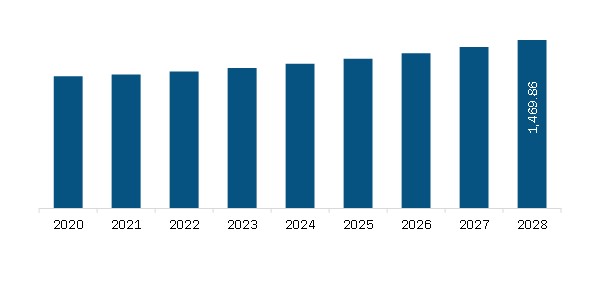
| Market size in 2021 | US$ 1,128.37 Million |
| Market Size by 2028 | US$ 1,469.86 Million |
| CAGR (2021 - 2028) | 3.8% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Type
|
|
Regions and Countries Covered
|
|
| Europe | UK, Germany, France, Russia, Italy, Rest of Europe |
| Market leaders and key company profiles |
|

The Europe Carbapenem-Based Antibiotics Market is valued at US$ 1,128.37 Million in 2021, it is projected to reach US$ 1,469.86 Million by 2028.
As per our report Europe Carbapenem-Based Antibiotics Market, the market size is valued at US$ 1,128.37 Million in 2021, projecting it to reach US$ 1,469.86 Million by 2028. This translates to a CAGR of approximately 3.8% during the forecast period.
The Europe Carbapenem-Based Antibiotics Market report typically cover these key segments-
The historic period, base year, and forecast period can vary slightly depending on the specific market research report. However, for the Europe Carbapenem-Based Antibiotics Market report:
The Europe Carbapenem-Based Antibiotics Market is populated by several key players, each contributing to its growth and innovation. Some of the major players include:
The Europe Carbapenem-Based Antibiotics Market report is valuable for diverse stakeholders, including:
Essentially, anyone involved in or considering involvement in the Europe Carbapenem-Based Antibiotics Market value chain can benefit from the information contained in a comprehensive market report.
Please tell us your area of interest
(Market Segments/ Regions and Countries/ Companies)