Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Forecast to 2028 - COVID-19 Impact and Regional Analysis By Organization Size (Small and Medium Organizations and Large Organizations), Offerings (Products, Platforms, and Services), Application (Product Design and Discovery, Product Development, Pre-Clinical Targeting, Assessment of Drugs and Other Biomedical Products, and Others), Clinical Indication (Cardiovascular Diseases, Neurodegenerative Diseases, Oncology, Rare Diseases, Metabolic Diseases, Immune Based Diseases, Infectious Diseases, and Others), and End User (Pharmaceutical and Biopharmaceutical Companies, Medical Technology Companies, Contract Research Organizations, and Others)
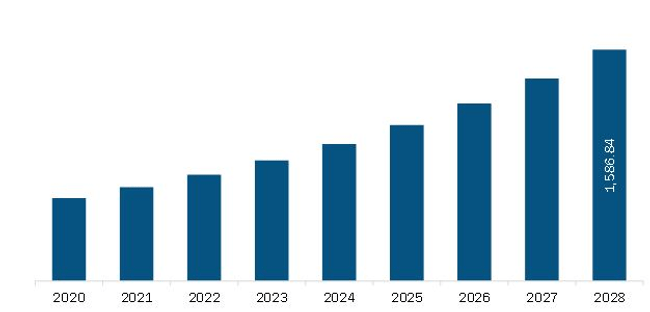
The Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is expected to reach US$ 1,586.8 million by 2028 from US$ 643.6 million in 2021. The market is estimated to grow at a CAGR of 13.8% from 2021 to 2028.
The rising application and need for in-silico trials in the orthopedic industry is a remarkable opportunity for key players operating in the in-silico trial market. The orthopedic device industry relies heavily on clinical evaluation to confirm its implants' safety, performance, and clinical benefits. In-silico clinical trial provides a viable technical and regulatory strategy for characterizing the full spectrum of patients, clinical conditions, and configurations embodied in modern orthopedic implant systems. Regulatory agencies are increasingly open to the use of computer-generated evidence. Therefore, in-silico clinical trials have the potential to become a new viable source for credible data on clinical device performance. Thus, rising opportunities in the development of orthopedic implants would support the in-silico trial market in the coming years.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market at a promising CAGR during the forecast period.
Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
Get more information on this report :
The market for in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into organization size, offerings, application, clinical indication, and end user. Based on organization size, the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into small and medium organizations and large organizations. Based on offerings, the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into products, platforms, and services. Based on application, the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into product design and discovery, product development, pre-clinical targeting, assessment of drugs and other biomedical products, and others. Based on clinical indication, the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into cardiovascular diseases, neurodegenerative diseases, oncology, rare diseases, metabolic diseases, immune based diseases, infectious diseases, and others. Based on end user, the in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into pharmaceutical and biopharmaceutical companies, medical technology companies, contract research organizations, and others. By country, the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market is segmented into China, Japan, India, Australia, South Korea, and the rest of Asia Pacific.
Dassault Systèmes SE; Certara Inc.; Ansys, Inc.; Sensyne Health plc.; and Cerner Corporation are among the leading companies in the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market.
1. Introduction
1.1 Scope of the Study
1.2 The Insight Partners Research Report Guidance
1.2.1 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By Organization Size
1.2.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By Offering
1.2.3 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By Application
1.2.4 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By Clinical Indication
1.2.5 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By End User
1.2.6 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – By Geography
2. Key Takeaways
3. Research Methodology
3.1 Coverage
3.2 Secondary Research
3.3 Primary Research
4. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Market Landscape
4.1 Overview
4.2 PEST Analysis
4.2.1 Asia Pacific PEST Analysis
4.3 Experts Opinion
5. In-Silico Trial Market – Key Market Dynamics
5.1 Market Drivers
5.1.1 Rising Concerns Over Animal Welfare and Benefits
5.1.2 Insufficient Variable Patient Data and Limited Timescale
5.2 Market Restraints
5.2.1 Insufficient Variable Patient Data and Limited Timescale
5.3 Market Opportunities
5.3.1 Growing Applications of In-silico Trials in Radiology and Orthopedic Device Industry
5.4 Future Trends
5.4.1 Adoption of Artificial Intelligence in In-Silico Drug Discovery
5.5 Impact Analysis
6. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market– Asia Pacific Analysis
6.1 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue Forecast and Analysis
7. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028– by Organization Size
7.1 Overview
7.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2020 & 2028 (%)
7.3 Large Organizations
7.3.1 Overview
7.3.2 Large Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts to 2028 (US$ Million)
7.4 Small and Medium Organizations
7.4.1 Overview
7.4.2 Small and Medium Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts to 2028 (US$ Million)
8. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 – Offering
8.1 Overview
8.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
8.3 Products
8.3.1 Overview
8.3.2 Products: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
8.4 Platforms
8.4.1 Overview
8.4.2 Platforms: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
8.5 Services
8.5.1 Overview
8.5.2 Services: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
9. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 – Application
9.1 Overview
9.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
9.3 Product Design and Discovery
9.3.1 Overview
9.3.2 Product Design and Discovery: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
9.4 Product Development
9.4.1 Overview
9.4.2 Product Development: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
9.5 Pre-Clinical Targeting
9.5.1 Overview
9.5.2 Pre-Clinical Targeting: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
9.6 Assessment of Drugs and Other Biomedical Products
9.6.1 Overview
9.6.2 Assessment of Drugs and Other Biomedical Products: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
9.7 Others (Optimization and Market Access)
9.7.1 Overview
9.7.2 Others (Optimization and Market Access): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 – Clinical Indication
10.1 Overview
10.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
10.3 Cardiovascular Disease
10.3.1 Overview
10.3.2 Cardiovascular Disease: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.4 Neurodegenerative Disease
10.4.1 Overview
10.4.2 Neurodegenerative Disease: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.5 Oncology
10.5.1 Overview
10.5.2 Oncology: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.6 Rare Diseases
10.6.1 Overview
10.6.2 Rare Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.7 Metabolic Diseases
10.7.1 Overview
10.7.2 Metabolic Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.8 Immune Based Diseases
10.8.1 Overview
10.8.2 Immune Based Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.9 Infectious Diseases
10.9.1 Overview
10.9.2 Infectious Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
10.10 Others (Haematology, Diabetes, Dermatology)
10.10.1 Overview
10.10.2 Others (Haematology, Diabetes, and Dermatology): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
11. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 – End User
11.1 Overview
11.2 Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
11.3 Pharmaceutical and Biotechnology Companies
11.3.1 Overview
11.3.2 Pharmaceutical and Biotechnology Companies: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
11.4 Medical Technology Companies
11.4.1 Overview
11.4.2 Medical Technology Companies: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
11.5 Contract Research Organizations
11.5.1 Overview
11.5.2 Contract Research Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
11.6 Others (Universities and Research Centres)
11.6.1 Overview
11.6.2 Others (Universities and Research Centres): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (US$ Million)
12. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts to 2028 – Geographical Analysis
12.1 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market
12.1.1 Overview
12.1.2 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market - Revenue and Forecast to 2028 (USD Million)
12.1.3 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.4 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.5 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.6 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.7 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8 Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Country, 2021 & 2028 (%)
12.1.8.1 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.1.1 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.1.2 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.1.3 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.1.4 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.1.5 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.1.6 China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8.2 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.2.1 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.2.2 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.2.3 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.2.4 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.2.5 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.2.6 Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8.3 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.3.1 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.3.2 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.3.3 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.3.4 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.3.5 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.3.6 India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8.4 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.4.1 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.4.2 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.4.3 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.4.4 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.4.5 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.4.6 Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8.5 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.5.1 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.5.2 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.5.3 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.5.4 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.5.5 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.5.6 South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
12.1.8.6 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.6.1 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
12.1.8.6.2 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size, 2019–2028 (USD Million)
12.1.8.6.3 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings, 2019–2028 (USD Million)
12.1.8.6.4 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application, 2019–2028 (USD Million)
12.1.8.6.5 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication, 2019–2028 (USD Million)
12.1.8.6.6 Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User, 2019–2028 (USD Million)
13. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market–Industry Landscape
13.1 Overview
13.2 Growth Strategies in the In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market (%)
13.3 Organic Developments
13.3.1 Overview
13.4 Inorganic Developments
13.4.1 Overview
14. Company Profiles
14.1 Dassault Systèmes SE
14.1.1 Key Facts
14.1.2 Business Description
14.1.3 Products and Services
14.1.4 Financial Overview
14.1.5 SWOT Analysis
14.1.6 Key Developments
14.2 Certara Inc.
14.2.1 Key Facts
14.2.2 Business Description
14.2.3 Products and Services
14.2.4 Financial Overview
14.2.5 SWOT Analysis
14.2.6 Key Developments
14.3 Ansys, Inc.
14.3.1 Key Facts
14.3.2 Business Description
14.3.3 Products and Services
14.3.4 Financial Overview
14.3.5 SWOT Analysis
14.3.6 Key Developments
14.4 Synopsys, Inc.
14.4.1 Key Facts
14.4.2 Business Description
14.4.3 Products and Services
14.4.4 Financial Overview
14.4.5 SWOT Analysis
14.4.6 Key Developments
14.5 Cerner Corporation
14.5.1 Key Facts
14.5.2 Business Description
14.5.3 Products and Services
14.5.4 Financial Overview
14.5.5 SWOT Analysis
14.5.6 Key Developments
15. Appendix
15.1 About The Insight Partners
15.2 Glossary of Terms
LIST OF TABLES
Table 1. Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 2. Asia Pacific In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 3. Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 4. Asia Pacific In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 5. Asia Pacific In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 6. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 7. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 8. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 9. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 10. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 11. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 12. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 13. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 14. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 15. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 16. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 17. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 18. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 19. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 20. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 21. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 22. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 23. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 24. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 25. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 26. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 27. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 28. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 29. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 30. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 31. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Organization Size – Revenue and Forecast to 2028 (USD Million)
Table 32. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Offerings – Revenue and Forecast to 2028 (USD Million)
Table 33. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, By Application – Revenue and Forecast to 2028 (USD Million)
Table 34. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Clinical Indication – Revenue and Forecast to 2028 (USD Million)
Table 35. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by End User– Revenue and Forecast to 2028 (USD Million)
Table 36. Organic Developments in the In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market
Table 37. Inorganic Developments in the In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market
Table 38. Glossary of Terms
LIST OF FIGURES
Figure 1. In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Segmentation
Figure 2. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Overview
Figure 3. Large Organizations Segment Held Largest Share of Organization Size Segment in In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market
Figure 4. India is Expected to Show Remarkable Growth During the Forecast Period
Figure 5. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, Industry Landscape
Figure 6. Asia Pacific: PEST Analysis
Figure 7. Experts Opinion
Figure 8. In-Silico Trial Market: Impact Analysis of Drivers and Restraints
Figure 9. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market– Revenue Forecast and Analysis – 2020- 2028
Figure 10. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Organization Size, 2020 & 2028 (%)
Figure 11. Large Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts to 2028 (US$ Million)
Figure 12. Small and Medium Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts to 2028 (US$ Million)
Figure 13. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
Figure 14. Products: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 15. Platforms: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 16. Services: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 17. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
Figure 18. Product Design and Discovery: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 19. Product Development: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 20. Pre-Clinical Targeting: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 21. Assessment of Drugs and Other Biomedical Products: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 22. Others (Optimization and Market Access): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 23. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
Figure 24. Cardiovascular Disease: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 25. Neurodegenerative Disease: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 26. Oncology: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 27. Rare Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 28. Metabolic Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 29. Immune Based Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 30. Infectious Diseases: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 31. Others (Hematology, Diabetes, and Dermatology): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 32. Asia Pacific In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Share by Segment - 2020 & 2028 (%)
Figure 33. Pharmaceutical and Biotechnology Companies: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 34. Medical Technology Companies: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 35. Contract Research Organizations: In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 36. Others (Universities and Research Centers): In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecasts To 2028 (US$ Million)
Figure 37. Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Key Country – Revenue (2021) (USD Million)
Figure 38. Asia Pacific In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market Revenue and Forecast to 2028 (USD Million)
Figure 39. Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market, by Country, 2021 & 2028 (%)
Figure 40. China: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 41. Japan: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 42. India: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 43. Australia: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 44. South Korea: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 45. Rest of Asia Pacific: In Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market – Revenue and Forecast to 2028 (USD Million)
Figure 46. Growth Strategies in the In-Silico Trials: Computational Modelling and Simulation for Medical Product Innovation and Regulatory Clearance Market (%)
- Dassault Systèmes SE
- Certara Inc.
- Ansys, Inc.
- Synopsys, Inc.
- Cerner Corporation
- Save and reduce time carrying out entry-level research by identifying the growth, size, leading players, and segments in the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market.
- Highlights key business priorities in order to assist companies to realign their business strategies.
- The key findings and recommendations highlight crucial progressive industry trends in the Asia Pacific in-silico trials: computational modelling and simulation for medical product innovation and regulatory clearance market, thereby allowing players across the value chain to develop effective long-term strategies.
- Develop/modify business expansion plans by using substantial growth offering developed and emerging markets.
- Scrutinize in-depth APAC market trends and outlook coupled with the factors driving the market, as well as those hindering it.
- Enhance the decision-making process by understanding the strategies that underpin security interest with respect to client products, segmentation, pricing and distribution.